July 12th, 2022
Written by: Marissa Maroni
Approximately 10% of adults aged 65 and older have Alzheimer’s disease (AD) in America in 20221. Alzheimer’s disease is a neurodegenerative disorder in which individuals struggle with difficulty remembering, depression, and impaired communication. The two cellular hallmarks of Alzheimer’s disease are abnormal protein deposits inside and outside of neurons. These proteins are made up of neurofibrillary protein tau tangles and amyloid-beta plaques, respectively. Despite being well studied in postmortem brains of patients, scientists still do not have a complete understanding for how these pathologies are created and formed early on in disease progression. This information could be key to developing treatments and a cure. Prior research has found a cellular cleanup process called autophagy is known to be dysregulated in AD but the mechanisms by which this happens is not fully understood. In a recent study, a group of scientists from New York University aimed to understand how defective autophagy could lead to the formation of amyloid-beta plaques2.
What is autophagy?
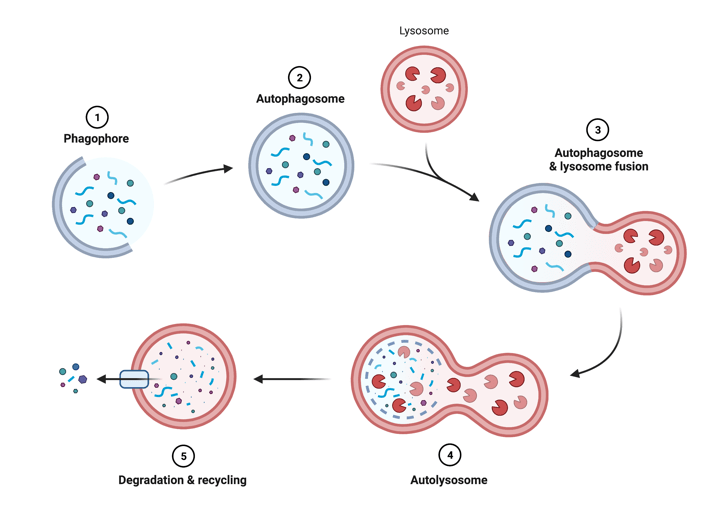
Autophagy is a process that breaks down unneeded proteins and debris in the cell3. Just like a city, cells in your body accumulate trash that needs to be disposed of. The process of cellular cleanup has two main players: an autophagosome which acts like a cell packager and a lysosome which acts like a trash incinerator. During this process, cellular debris is engulfed into an autophagosome (packager) that fuses with a lysosome (trash incinerator). Once together, the digestive enzymes in the lysosome break down debris which can then be released and recycled (figure 1).
Experimental setup: Using acidity to examine autophagy function
The scientists wanted to examine the process of autophagy in the brain to be able to determine what may be defective in an AD brain (see figure 1). To test this, they needed to be able to visualize the autophagosomes/autolysosomes. They used a genetically altered mouse model that expresses a protein found in autophagosomes/autolysosomes that fluoresces so that they can see the protein under a microscope. This fluorescent marker is unique because not only does it mark autophagosomes and autolysosomes, but it also changes color depending on the acidity. This feature is incredibly useful for understanding autolysosome function as a properly functioning autolysosome will acidify after formation to activate enzymes that break down material (see stage 4 in figure 1). Using this tool, they can monitor the autophagy process and determine if it is dysfunctional based on the PH indicator marker. This mouse model was then crossed with mice that develop amyloid-beta plaques in the brain, recapitulating a hallmark of AD pathology, so that the offspring had amyloid-beta plaques and their autophagosomes/autolysosomes could be visualized. Together, this model can help researchers assess how the process of autophagy is impacted during AD.
Autolysosome dysfunction and AD-related proteins accumulate
Using their mouse model, the researchers were able to image the brains of AD and control mice and quantify the number and size of their autolysosomes. They found that there was an increase in autolysosomes that were poorly acidified in the AD mouse model compared to controls. Remember lysosomes are like trash incinerators and in this case the poorly acidified autolysosomes are the equivalent of a faulty trash incinerator that’s unable to completely degrade the engulfed material. This finding demonstrates that in AD the defects in autophagy could be due to poor degradation of the engulfed material. The researchers wanted to see if this dysfunction in autophagy could be tied to AD. To do this, they examined the aptly named protein, amyloid precursor protein (APP) which is a larger protein that is cut to make amyloid-beta and they examined amyloid-beta itself. Using a technique called immunostaining, researchers were able to mark areas in the cells that had APP and amyloid-beta. They found that APP and amyloid-beta accumulated most in the poorly acidified autolysosomes in the AD mice. Together, this research indicates that autolysosomes are dysfunctional in mouse models of AD and that APP and amyloid-beta accumulate in them.
Autolysosomal stress forms flower-like structure that turn into amyloid-beta plaques
Based on prior findings the scientists hypothesized that these dysfunctional autolysosomes in neurons may accumulate APP and amyloid-beta and create amyloid plaques. Amyloid plaques are created after a neuron filled with amyloid-beta protein dies where the amyloid-beta aggregates to form the plaques4. To test whether amyloid plaques are created from these neurons, the scientists examined the dysfunctional autolysosomes over time. Interestingly, they found using that the autolysosomes swelled and formed a unique structure similar to the shape of a flower around the nucleus of the neuron. Using imaging techniques, they found that amyloid-beta filled neurons with autophagy dysfunction would eventually die and leave in its wake amyloid-beta plaques. This finding connects autophagy dysfunction to AD and amyloid-beta plaque formation. To connect this back to our metaphor, imagine each neuron in your brain as its own miniature city. In a diseased neuron, the city’s waste management is dysfunctional (autolysosomes) and continues to aggregate toxic waste causing the city to swell with trash (amyloid-beta). Eventually, the city combusts and leaves behind a ghost of its waste in its wake (amyloid-beta plaques).
Research studies such as this take huge steps towards understanding AD and being able to better treat individuals with this disease. In the future, researchers could potentially monitor autophagy function and work on ways to improve autophagy in AD patients to improve patient outcomes!
References
1. Alzheimer’s Association. (2022) 2022 Alzheimer’s Disease Facts and Figures. Alzheimers Dement 2022;18.
2. Lee, J. H., Yang, D. S., Goulbourne, C. N., Im, E., Stavrides, P., Pensalfini, A., … & Nixon, R. A. (2022). Faulty autolysosome acidification in Alzheimer’s disease mouse models induces autophagic build-up of Aβ in neurons, yielding senile plaques. Nature Neuroscience, 1-14.
3. Mizushima, N. (2007). Autophagy: process and function. Genes & development, 21(22), 2861-2873.
4. Takahashi, R. H., Nagao, T., & Gouras, G. K. (2017). Plaque formation and the intraneuronal accumulation of β‐amyloid in Alzheimer’s disease. Pathology international, 67(4), 185-193.
Figure 1 created in BioRender
Cover photo by Colin Behrens from Pixabay