February 18, 2020
Written by: Nitsan Goldstein for IAES

One of the brain’s most mysterious functions is the formation and storage of memories. In the last fifty years or so, however, neuroscientists have made immense strides in understanding what might go into forming a memory. We therefore have a better handle on how diseases such as autoimmune encephalitis (AE) impair memory function. Here we will discuss what goes into creating a memory, how cells in the brain are built to form lasting connections with each other, and why the processes underlying memory are particularly vulnerable in AE.
Step-by-step memory formation
We are constantly taking in information about the world around us. Only a subset of this information, however, becomes a memory. Uncovering the cellular processes underlying this phenomenon have been critical in our understanding of how memory works in the brain. The hippocampus is an essential brain region for memory. The hippocampus is a kind of hub of information, and is necessary for forming new memories and making connections between different kinds of sensory experiences. For example, if you’re walking down the street and notice a pleasant smell from a new bakery, your hippocampus will associate that smell with your location and the image of the bakery. If you’re paying close enough attention, the information will be stored and recalled the next time you want a pastry. How are these connections made?
Memory formation involves long term changes in neuronal communication
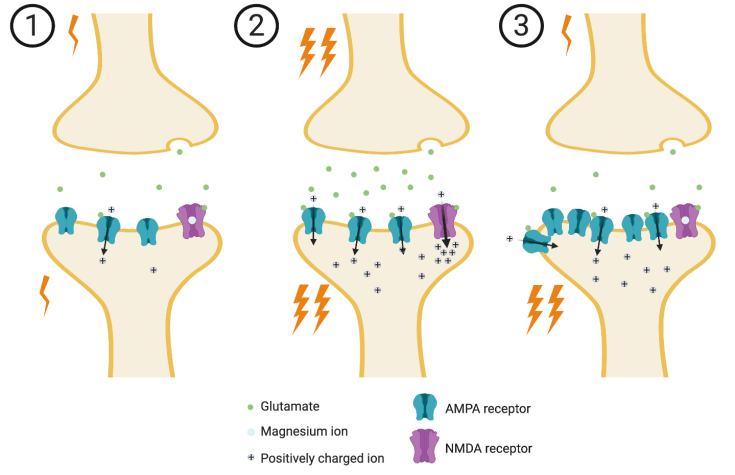
It is believed that memory formation relies on a very simple principle in neuroscience: frequent communication between two neurons strengthens the connections between those neurons. Neurons in the hippocampus and other areas in the brain communicate with each other using a molecule called glutamate, which is released into the tiny spaces between neurons, which are called synapses. Glutamate is released when a neuron (the pre-synaptic neuron) becomes active and it binds to glutamate receptors on the neuron it is communicating with (the post-synaptic neuron). The binding of glutamate to its receptor typically causes the post-synaptic neuron to become active, and thus the signal is transmitted from one neuron to the next.
The effect of glutamate on the post-synaptic neuron can be weakened or strengthened, depending on how strongly and frequently the pre- and post-synaptic neurons are simultaneously active. For example, if there is a strong, sustained signal from the pre-synaptic neuron that activates the post-synaptic neuron, the two neurons will be simultaneously active. When this happens, the synapse is altered such that glutamate strongly activates the post-synaptic neuron. If the signal from the pre-synaptic neuron is small and brief, the neurons will not be simultaneously active. Thus, the post-synaptic response to glutamate is weak.
The glutamate receptors are critical for strengthening or weakening communication between two neurons (see Figure 1). There are two types of glutamate receptors at play: AMPA receptors and NMDA receptors1. Most of the signal is transmitted through AMPA receptors. When glutamate binds to them, they open like a gate to let ions into the neuron. Ions are molecules that have a positive or negative electrical charge, like a battery. The AMPA receptor gates let positively charged ions through, which activates the neuron. NMDA receptors, on the other hand, typically do not let ions in because they have a magnesium ion blocking the gate. When the pre-synaptic neuron is strongly activated, a lot of glutamate binds to the AMPA receptors, causing an influx of positive ions in the post-synaptic neuron. When enough positive ions enter the neuron through the AMPA receptors, the magnesium ion in the NMDA receptor is kicked out, allowing a rush of even more positive ions through its gates. Once this happens, many long-term changes can occur inside the neuron. For example, more AMPA receptors are shuttled to the cell membrane1. Such changes ensure that the next time the pre-synaptic neuron releases glutamate, the response from the post-synaptic neuron will be larger. It is through these processes that sensations and experiences are represented in the hippocampus as groups of neurons that are strongly activated together. The solidification of these groups and the signals they send to other areas of the brain underlie memory formation.
Memory in Autoimmune Encephalitis
Autoimmune encephalitis occurs when antibodies targeting brain cells penetrate the brain. Memory impairments are a common symptom of AE2. This is especially true of cases where the body generates antibodies that target receptors involved in learning and memory, such as AMPA and NMDA receptors. Scientists in recent years have used animal models to study what exactly the antibodies are doing to the receptors in the brain. One group, for example, took serum from the blood of patients that had AE with antibodies against the AMPA receptor3. They then infused the serum into the brains of healthy mice. They found that the mice had more difficulty remembering objects they had encountered previously compared to mice that received infusions of serum from healthy control subjects. When they looked at the hippocampus, they found that the neurons had fewer AMPA receptors. Through further studies they determined that the binding of the antibody to the AMPA receptor causes the neuron to move the receptor from the membrane to the inside of the neuron, such that glutamate is no longer able to bind it (see Figure 2).

Other studies have shown similar effects from serum taken from patients with AE containing antibodies against the NMDA receptor. Mice whose brains were exposed to this type of serum show deficits in learning and memory, antibody accumulation in the hippocampus, and fewer NMDA receptors on the surface of neurons4. Even antibodies targeting other proteins, such as LGI1, can affect memory processes. LGI1 is a protein that helps link the pre- and post-synaptic neurons together, stabilizing the synapse. In this case, the antibodies decrease the number of AMPA receptors, preventing proper communication between the pre- and post-synaptic neurons in the hippocampus and other regions of the brain5,6.
Future Work
While most patients fully recover from memory impairments with encephalitis treatment, memory loss may persist longer than other symptoms. More work is needed to understand what kind of long-term changes may occur in the hippocampus that would explain prolonged memory deficits. It’s possible that the antibodies actually cause irreversible neuron death in the hippocampus7. Alternatively, the neurons may undergo changes to try to compensate for the lost AMPA or NMDA receptors3,7. These changes may take longer to reverse than other cellular processes that recover quickly following treatment.
References
- Luscher, C., Malenka, R.C. NMDA Receptor-Dependent Long-Term Potentiation and Long-Term Depression (LTP/LTD). Cold Spring Harb. Prospect. Biol. 4, a005710 (2012).
- Lancaster, E. The Diagnosis and Treatment of Autoimmune Encephalitis. J. Clin. Neurol. 12, 1-13 (2016).
- Haselmann, H., et al. Human Autoantibodies against the AMPA Receptor Subunit GluA2 Induce Receptor Reorganization and Memory Dysfunction. Neuron 100, 91-105 (2018).
- Planaguma, J., et al. Human N-methyl D-aspartate receptor antibodies alter memory and behaviour in mice. Brain 138, 94-109 (2014).
- Dalmau, J, & Graus, F. Antibody-Mediated Encephalitis. N. Engl. J. Med. 378, 840-851 (2018).
- Ohkawa, T. et al. Autoantibodies to epilepsy-related LGI1 in Limbic Encephalitis neutralize LGI1-ADAM22 interaction and reduce synaptic AMPA receptors. J. Neurosci. 33, 18161-18174 (2013).
- Moscato, E.H., Jain, A., Peng, X., Hughes, E.G., Dalmau, J., & Balice-Gordon, R.J. Mechanisms underlying autoimmune synaptic encephalitis leading to disorders of memory, behavior and cognition: insights from molecular, cellular and synaptic studies. Eur. J. Neurosci. 32, 298-309 (2010).
Images
Cover image via Pixabay
Figures 1 & 2 created using BioRender
