March 28th, 2023
Written by: Stephen Wisser
Buying a car can be a painful process. With so many options, how do you choose the best car that fits your needs? Thankfully, there are many resources available to help you find that perfect vehicle. There are countless websites that review cars, entire magazines devoted to cars, stores that sell just cars, and of course, friends and family that can tell you about their cars. With all these research options and a little patience, you should have all the information available to narrow down your search to a few cars, test drive some, and then make a final decision.
Unfortunately, as neuroscientists (and really all scientists for that matter), we aren’t so lucky in our day-to-day lives to have all this information available. Afterall, the goal of science is to ask questions and discover previously unknown things. So how do we decide which ideas to “test drive” in the lab and where to focus our experiments? We often start in a similar place as someone looking to buy a car except in our case, we are mostly looking for a specific region in the brain to focus on. We search through the literature and ask colleagues before deciding on a few “cars to test drive” or candidate brain regions to investigate. This becomes particularly difficult if we want to do something entirely new, like see the effects of a new drug on brain activity. A good first pass at searching the brain for these candidate regions could be to use a technique known as magnetic resonance imaging, or MRI, which is a common method that doctors use to scan human brains. Unfortunately, MRI scans are like a blurry picture and don’t allow us to see brain activity in great detail, a technical term called spatial resolution. The spatial resolution of MRIs is very low, such that each pixel on a MRI picture measures the activity of millions of neurons.1 That would be like describing 1 million people with a single word! We are clearly missing important information about brain activity in MRI scans since we can’t see individual neurons. We need a more precise way to identify active brain regions to test drive.
With the invention of two newer technologies, called optical clearing and lightsheet microscopy, scanning the whole brain for active regions at the level of individual neurons is now a reality. It is important to note that for practical and ethical reasons, these techniques are only done in animal models, which can and do tend to carry over well to humans. These new techniques make the process of scanning whole animal brains quick and efficient, bringing scientists closer to that car customer who has all the information to decide which vehicle to test drive.
Technology 1: Optical Clearing
So what are these two techniques and how do they work? Let’s start with the first: optical clearing. Maybe in your early school years, you had the chance to look at some simple things, like a leaf, under a microscope. This microscope likely used a light bulb to shine light through the leaf and allow you to see it nice and bright through the eyepiece. This works well for things that are thin because the light can easily pass through it. Something thicker, like a whole worm or tadpole, can’t be seen as well under the microscope because it is so thick that the light can’t pass through it. The same is true for much larger and fancier microscopes. Putting a whole mouse brain under a microscope (yes, they are actually decently large) will block all the light and you won’t see anything. Scientists normally have to cut the brain into hundreds of thin sections that light can pass through using what is essentially a small deli meat slicer. They then look at each slice one by one – a dreadfully long process. Optical clearing protocols offer an alternative to this slicing method by chemically treating the whole brain so that it becomes clear. This allows light to pass through it so that it can be seen under a microscope.
There are many different optical clearing protocols, but the science for all of them is roughly the same. Using detergents similar to the ones found in your shampoo, the fats in the brains are first washed out. Fats are the main component of brains that are responsible for scattering light so that it can’t pass through. The brains are typically washed in this detergent solution for several days and after some other washing steps are ready to be imaged. The whole procedure can take as long as a month, and the brains are fully translucent in the end. This means that the entire brain, once cleared, can be placed intact into a microscope without any cutting and be imaged whole.
There are two main types of optical clearing protocols in use today, organic-based and aqueous (water) based that each offer different advantages. Since the differences between the 2 are technical, in this post I’ll focus on just one organic-based method known as “immunolabeling-enabled three-dimensional imaging of solvent-cleared organs” which most people just call iDISCO. This popular iDISCO protocol2 uses cheap and easily accessible chemicals in a simple recipe that clears brains quickly. Importantly, the procedure can be combined with a “staining” protocol that allows neuroscientists to look at neurons that have the protein cFos. This cFos protein is made by cells that were just active prior to collection of the brain tissue, so staining it is a good way to tell which neurons were active. The best part about the stain is that it glows, meaning the neurons that were active will glow in a microscope.
Technology 2: Lightsheet Microscopy
But how can we see these glowing neurons throughout the brain? This is where the second technology comes in to play – lightsheet microscopy. As the name implies, this microscope uses a sheet of laser light to light up the brain which can be seen through a lens. The brain is then moved up and down so that this sheet of laser light hits every part of the brain, taking a picture of each part or plane as it goes (figure 1). These pictures can then be stacked on top of each other to create a 3D image of the whole brain showing the once active neurons glowing in 3D space.
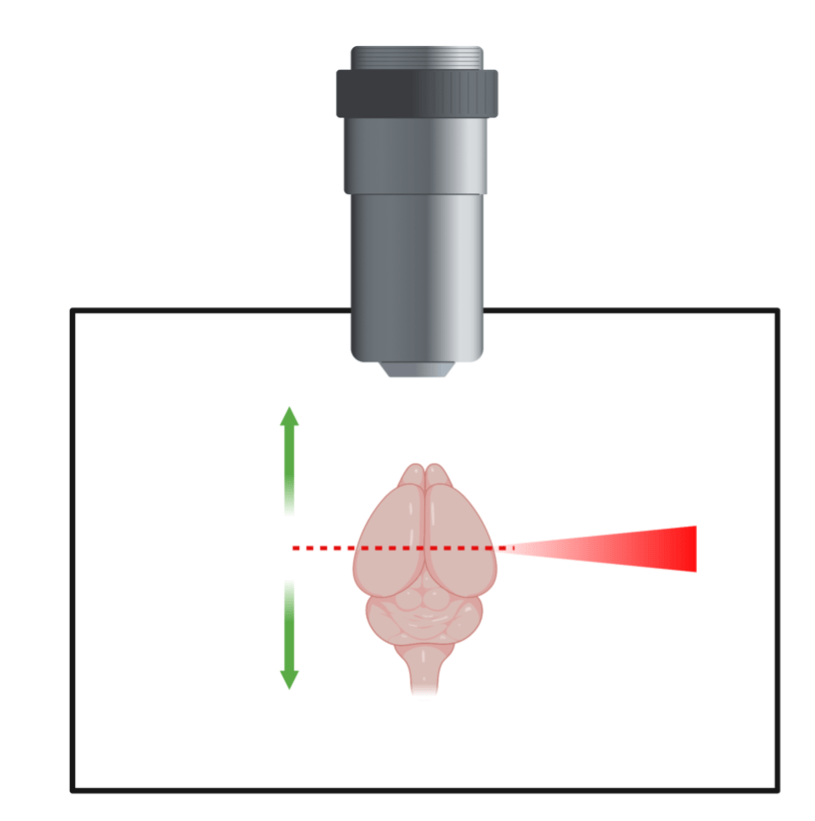
Once imaged, the glowing (active) neurons need to be counted. In a whole mouse brain, there can be tens of thousands of active neurons, so the field has adopted computational approaches to count the cells in each brain region. Using a computer program, the different regions in the brain are determined, much like state lines being drawn on a blank map of the US. Once the regions are identified, the neurons which appear as glowing bright dots are counted. In the end, a spreadsheet is spit out with the number of glowing, and thus active, neurons in each brain region. Using these spreadsheets, a scientist can analyze activity of individual neurons throughout the whole brain and get a clearer picture of which regions are active and worth looking into further.
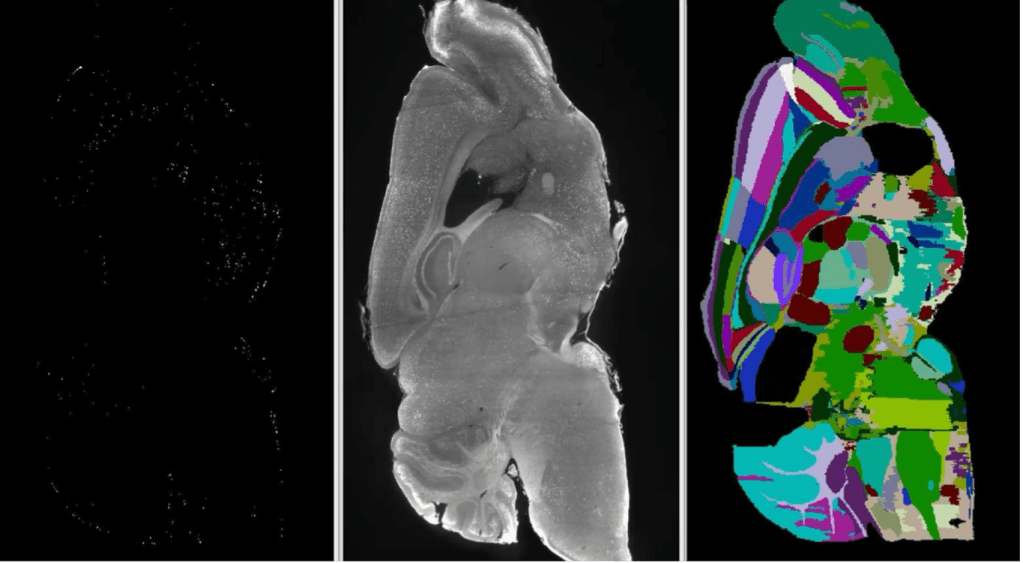
As someone who has worked for the last two years optimizing an iDISCO clearing and analysis process, I can tell you the process is not as straight forward as it may sound. There are many quality control steps that require a human’s judgement to move forward, and since people are different, results from this process can vary slightly depending on who’s doing the experiments. For this reason, such experiments should only be viewed as a first search of the brain to inform more experiments that need to be done. Remember, this process is just providing information on where to focus attention. Just like cars still need to be test driven before a final decision is made, candidate brain regions need to be further investigated. Now that the protocols and technology are mostly worked out, they are proving to be valuable especially in areas that are relatively newer scientific fields such as psychedelics. A recent publication used optical clearing and lightsheet microscopy to identify brain regions that were active in response to psilocybin (main ingredient in magic mushrooms) and ketamine3. This screener experiment will likely serve as a valuable starting point for more experiments in this emerging field that want to understand the mechanisms of these unique psychedelic compounds and how they might be used as new medicines to treat those with PTSD or depression. While not perfect, we now have something closer to what the car industry has in terms of information and can be more confident that we’re focusing on the absolute best brain regions moving forward.
References
1.Yuhas, Daisy. “What’s a Voxel and What Can It Tell Us? A Primer on Fmri.” Scientific American Blog Network, Scientific American, 21 June 2012, https://blogs.scientificamerican.com/observations/whats-a-voxel-and-what-can-it-tell-us-a-primer-on-fmri/.
2.Renier, Nicolas, et al. “iDISCO: a simple, rapid method to immunolabel large tissue samples for volume imaging.” Cell 159.4 (2014): 896-910.
3.Davoudian, Pasha A., Ling-Xiao Shao, and Alex C. Kwan. “Shared and distinct brain regions targeted for immediate early gene expression by ketamine and psilocybin.” ACS Chemical Neuroscience 14.3 (2023): 468-480.
4. Roy, Snehashis. “CATNIP: Whole Brain Cellular AcTivity Estimation : a Nice Image Processing Program.” Github, 12 Feb. 2023, https://github.com/snehashis-roy/CATNIP.
Cover photo by mohamed_hassan from Pixabay
Figure 1 made with BioRender.com
Figure 2 posted with permission of Dr. Snehashis Roy.
