October 25th, 2022
Written by: Kara McGaughey
We have all been taunted with the age-old adage, “You are what you eat.” In response, maybe you rolled your eyes while settling a fifth double-stuffed Halloween oreo onto your plate, knowing that — no matter how many you eat this Fall — you are not an oreo. There is, however, more truth to the sentiment than you might think, and it’s inspiring an emerging line of scientific work with the potential to transform our understanding of both brain health and disease.
How are the gut and the brain related?
Just as your brain and your spinal cord are part of your body’s central nervous system (CNS), your stomach and your bowels have their own nervous system called the enteric nervous system (ENS).1 While scientists used to think of the ENS as separate from the CNS, it turns out that the two nervous systems have quite a lot in common (Read more about the ENS in this previous PennNeuroKnow post!). For example, the ENS relies on the same type of neurons and neurotransmitters found in the brain. In fact, 95% of the neurotransmitter serotonin is produced in the gastrointestinal tract where it’s taken in through consumption of protein-based foods and later delivered to the brain.2 In addition to neurons and neurotransmitters, the gut contains a host of organisms (think: bacteria, viruses, fungi, and other microorganisms that live in the digestive tract), which scientists call microbes or microbiota. These microbiota are part of a bidirectional system called the gut-brain axis (arrows in Figure 1) that enables the ENS to communicate with the CNS and vice versa.1 While the routes of communication between gut microbiota and the brain are not fully understood, scientists are beginning to understand the myriad of ways in which this crosstalk may influence our health.
What does bidirectional communication between the gut and the brain mean for health and disease?
The ongoing dialogue between the ENS and the CNS has a host of consequences. Most of us have had firsthand experience with the unpleasant ways that negative emotions and stress can perturb gut function. The gut-brain axis is relevant not only for these moment-to-moment or day-to-day changes in brain state, but also for longer-lasting states of brain disorder and dysfunction. For example, a study of more than 18,000 subjects revealed that individuals with gastrointestinal symptoms have higher rates of mood and anxiety disorders.3 More broadly, ENS symptoms are now reported to accompany an increasing number of CNS disorders, ranging from neuropsychiatric (like depression or anxiety) to neurodegenerative (such as Alzheimer’s or Parkinson’s disease).3-4 This relationship between gastrointestinal dysfunction and brain-related disorders points to the gut-brain axis as not only a potential culprit for the transfer of disease between the CNS and ENS, but also as a potential avenue for treatment.
Could gut microbiota function as a treatment target?
The idea of using gut microbiota to treat disease and dysfunction dates back ~2,000 years to a Chinese medical technique called fecal microbiota transplant (FMT). During FMT, healthy bacteria or microbiota are extracted from the feces of a donor and transferred to a recipient. Similar to a kidney or liver transplant, the goal is to restore normal function. In this case, FMT aims to restore normal composition and function of the microbial community by replacing the recipient’s unhealthy gastrointestinal microbiota with the donor’s healthy microbiota. The transfer itself can take several forms: a nasogastric route (the insertion of a plastic tube through the nose, past the throat, and down into the stomach), enemas, or pills taken orally. While it may sound a lot like science fiction and a little gross, FMT is a quick, safe, and effective way of modifying the intestinal microbiota. It is already an FDA-approved treatment for an infection called Clostridium difficile, or C. diff, which occurs when an overgrowth of “bad” bacteria in the colon wreaks havoc on the digestive system. FMT replenishes healthy microbiota in C. diff patients, boasting cure rates of up to 90%.5-6
Could replacement of gut microbiota function as a treatment for brain-related disease and disorder?
It’s not just infections in the gastrointestinal system that can cause imbalances in the composition of gut microbiota. Recent work at the intersection of psychiatry, neuroscience, and microbiology has revealed that depression, anxiety, schizophrenia, Alzheimer’s disease, and a host of other brain-based pathologies are also associated with changes in gut microbiota (Figure 1).7-8 For instance, researchers compared the gut microbiota of patients with Parkinson’s disease with healthy control subjects and found some substantial differences. Critically, this comparison not only revealed differences in gut microbiota between subjects with and without Parkinson’s disease, but also differences in microbial composition between Parkinson’s patients. For example, patients with more difficulty walking and maintaining their posture had more of a particular microbe than patients with less severe symptoms.7 This relationship between disease presence, disease severity, and gut microbiota suggests that changes in microbial composition might be directly related to brain dysfunction.
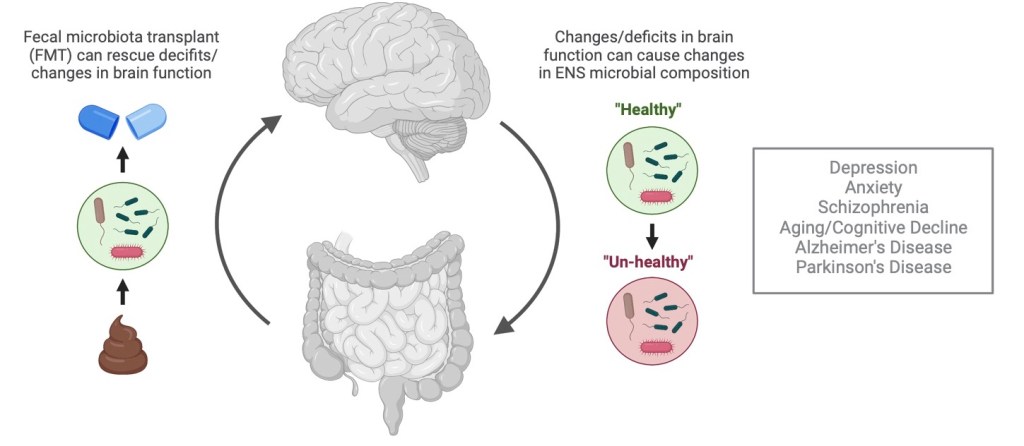
To explore if gut microbiota can cause and control the development of brain-related diseases and disorders, scientists collected fecal samples from a group of patients with severe depression and transferred the unhealthy bacteria to rats. After fecal microbiota transplant (FMT), the rats displayed behavioral and physiological features characteristic of depression.7 The same finding resulted from a study that used FMT to transfer microbiota from patients with schizophrenia into rodents. Not only did the rodents take on the unhealthy structure of the schizophrenia-related microbiome, but they also adopted schizophrenia-related behaviors.8Critically, not only can the transfer of disease-related microbiota via FMT produce disease-related behaviors, but FMT can use healthy microbiota to treat disease symptoms (Figure 1). In one study published last year, researchers used a stress paradigm to induce depressive-like behaviors in rats (a common animal model for stress-related disorders, like depression). They then took fecal material from control rats and used it to treat the depressed animals for 14 days via FMT. The microbiota transplant was so successful at reducing the depressive-like behaviors that there were no longer behavioral differences between the stressed and the control animals.9
Taken together, these results highlight the fact that microbiota can impact brain function and behavior. As such, the reorganization of microbial communities presents an exciting avenue for treating brain disorders and dysfunction, suggesting FMT as an appealing treatment option.
Fecal microbiota transplants can open many avenues for treatment, but should we pursue them?
Many advances have been made and the emerging research presents a promising outlook for the use of microbiota-targeted interventions for the treatment of brain-related diseases and disorders like depression. However, it is unlikely that FMT will replace traditional pharmacological approaches any time soon.
Why? Animal studies are establishing a basis for clinical trials and future work, but human-specific studies lag behind. Given the complex, interconnected nature of the gut-brain axis, transplanting microbial communities from person to person has the potential for major consequences, meaning that all human FMT donors must undergo time-intensive screening to avoid transfer of antibiotic-resistant organisms, viruses, and other pathogens. For example, in March of 2020, the FDA put out a buletin cautioning against the transfer of COVID-19 via FMT and mandating that donors (and their stool) undergo additional testing.10 Nonetheless, research in this area is continuing to take off, necessitating constant redefining of the boundaries between bodily systems, like the ENS and the CNS. Perhaps, you’ll now think twice when you hear “you are what you eat,” recognizing the interconnectedness of our gut and our brain and the treatment potential this connection unlocks.
Want to know more about microbiota and the gut-brain axis? You can check out previous PennNeuroKnow articles highlighting the role of microbiota in health and disease and brain development.
References
- Rao, M. & Gershon, M.D. The bowel and beyond: The enteric nervous system in neurological disorders. Nat. Rev. Gastroenterology & Hepatology. 13, 517-528 (2016).
- Shine, J. M., O’Callaghan, C., Walpola, I. C., Wainstein, G., Taylor, N., Aru, J., Huebner, B., & John, Y. J. Understanding the effects of serotonin in the brain through its role in the gastrointestinal tract. Brain. 9, 2967-2981 (2022).
- Walker, E. A., Katon, W. J., Jemelka, R. P., & Roy-Byrne, P. P. Comorbidity of gastrointestinal complaints, depression, and anxiety in the epidemiologic catchment area (ECA) study. American Journal of Medicine. 82, S26-S30 (1992).
- Madison, A. & Kiecolt-Glaser, J. K. Stress, depression, diet, and the gut microbiota: Human–bacteria interactions at the core of psychoneuroimmunology and nutrition. Current Opinion in Behavioral Sciences. 28, 105-110 (2019).
- Green J., Castle D., Berk M., Hair, C., Loughman, A., Nierenberg, A., Athan, E., & Jacka, F. Faecal microbiota transplants for depression – Who gives a crapsule? Aus N Z J Psychiatry. 53, 732-734 (2019).
- Ser, H-L., Letchumanan, V., Goh, B-H., Wong, S. H., & Lee, L-H. The use of fecal microbiome transplant in treating human diseases: Too early for poop? Frontiers in Microbiology. 12. (2021).
- Dinan, T. G. & Cryan, J. F. Gut feelings on Parkinson’s and depression. Cerebrum. 4-17 (2017).
- Zhu, F., Guo, R., Wang, W., Ju, Y., Wang, Q., et al. Transplantation of microbiota from drug-free patients with schizophrenia causes schizophrenia-like abnormal behaviors and dysregulated kynurenine metabolism in mice. Molecular Psychiatry. 25, 2905-2918 (2020).
- Rao, J., Qiao, Y., Xie, R., Lin, Y., Jiang, J., Wang, C., & Li, G. Fecal microbiota transplantation ameliorates stress-induced depression-like behaviors associated with the inhibition of glial and NLRP3 inflammasome in rat brain. Journal of Psychiatric Research. 137, 147-157 (2021).
- U.S. Food and Drug Administration. Safety alert regarding use of fecal microbiota for transplantation and additional safety protections pertaining to SARS-CoV-2 and COVID-19. https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/safety-alert-regarding-use-fecal-microbiota-transplantation-and-additional-safety-protections (2020).
Cover photo by Derek Owens on Unsplash.
Figure 1 made with Biorender.com.
