June 23, 2020
Written by: Yarden Wiesenfeld
This summer we’re excited to introduce some new Guest Authors at PennNeuroKnow! This week’s article come from Yarden Wiesenfeld, a Penn undergraduate student in Dr. Michael Granato’s lab who studies the molecular mechanisms that drive learning in zebrafish. She is a dancer in Penn’s swing dancing troupe and also loves art and cooking.
It is well-known that a mother’s caregiving instincts are rooted in biology. The female brain is re-wired during and after pregnancy to boost nurturing behavior. Signals originating in a region of the brain called the medial pre-optic area are involved in increasing reward when mothers interact with their babies1. Another part of the brain called the amygdala makes a mother more sensitive to her baby’s cries2. High levels of oxytocin, a hormone involved in social bonding, lead to greater motivation for maternal care3. And of course, a woman’s body must undergo physical and hormonal changes to breastfeed her baby.
But what about fathers? Only 3-5% of mammalian species are biparental, in which fathers engage in direct caregiving, such as grooming or carrying offspring, outside of paternal behavior like guarding and providing food4. Thus, there is much to be learned about how paternal investment shapes a man’s brain and what impact it has on the growing child. As the role of fathers in caregiving has increased in the Western world over the past few decades, there has been greater interest in understanding the neurobiological changes that are involved in fatherhood.
Fathers actually undergo many of the hormonal changes that are seen in mothers. Prolactin, the hormone that tells a woman’s body to make breastmilk, increases in emotionally involved fathers, too. Experienced fathers show elevated prolactin levels when they hear a baby crying5. Additionally, oxytocin levels are similar in mothers and fathers6. Similar to mothers, oxytocin becomes elevated in fathers through physical contact with their baby, such as skin-to-skin, and diminishes less quickly than for mothers7. Nasal administration of oxytocin causes both men and women to be more attuned to their baby8,9. Furthermore, in primary-caregiving heterosexual mothers and primary-caregiving homosexual fathers (no maternal involvement), there is a relationship between oxytocin levels of the parent and the child. High oxytocin in a parent during their child’s infancy leads to enhanced connectivity of neural networks linked to caregiving, which can predict their child’s oxytocin levels and social development as far as preschool6. Finally, testosterone declines sharply in men when they have a baby. Interestingly, the hormone decreases in fathers as they become more involved in caring for their child10.
Despite the similarities in hormonal changes, the neural networks involved in caregiving behaviors may vary between mothers and fathers. While these networks are not mutually exclusive, scientists believe that maternal care is largely subcortical and hormonally regulated, while paternal care is mainly socio-cognitive and based on experience.
The maternal pathway: Subcortical structures, like the amygdala and motivation-reward limbic network, promote an emotional attachment to a baby’s needs and physical pain. Vigilance for the child’s wellbeing and reward from the mom-baby bond are supported by this network and are modulated by pregnancy hormones, like oxytocin and prolactin. This subcortical circuitry seems stronger in mothers than in fathers; amygdala activation in response to infant crying is five times higher in mothers than in secondary-caregiving fathers. The subcortical parenting network is an ancient system, providing nonconscious motivation for care in other mammals from rodents to primates. However, the human caregiving network is connected to later-evolving cortical regions that define the human ‘social brain,’ which provides the conscious ability to infer what our babies are feeling and adapt to their nonverbal signals11.
The paternal pathway: With experience, cortical networks develop to support interpretation of infant signals (mentalization), synchrony between the actions of child and parent (embodied simulation/mirror), and executive control and multitasking (emotion regulation). The ‘mirror’ network, thought to be involved in empathy, helps parents to represent the actions and goals of their baby in their own brain. This network is consistently activated in all father studies, but not all mother studies. Parallel to oxytocin in mothers, the male bonding hormone vasopressin is correlated with amygdala activation in fathers11.
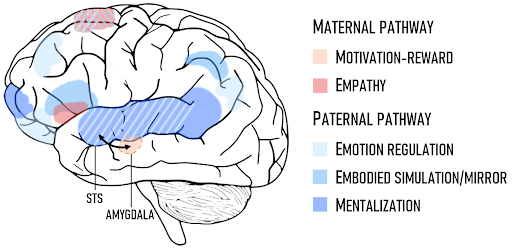
Are these differences due to sex, or a parent’s traditional role in caregiving? Interestingly, studies of primary-caregiving homosexual fathers indicate an interlinking of the two pathways. Primary-caregiving fathers showed activation in the superior temporal sulcus (STS), a region that helps parents to represent their baby’s mental state (mentalization), that matched secondary-caregiving fathers, and high amygdala activation similar to mothers. Furthermore, only the primary-caregiving fathers demonstrated functional connectivity between the amygdala and STS. Researchers believe this indicates a recruitment of the maternal pathway in men in the absence of a mother. This interregional wiring demonstrates enhanced neural plasticity, or “malleability” of the brain. Connections between the amygdala and STS increased with the amount of time spent caregiving. Thus, a more prominent role in parenting is accommodated by a greater ability for a parent’s brain networks to be rejiggered in response to infant inputs12.
We can learn something valuable from this research — that optimal parenting involves the entire range of ways of bonding with one’s child, and that the ability to engage caregiving networks is dependent not on gender, but on caregiving role. Our long-held ideas of what parenting looks like restricts our understanding of human caregiving.
So, dads, we want to thank you for your care. For adapting to our behaviors, for helping to develop our social brains, and for allowing us to “change your mind,” even if you didn’t know it was happening.
References:
- Numan, M. & Stolzenberg, D. S. Medial preoptic area interactions with dopamine neural systems in the control of the onset and maintenance of maternal behavior in rats. Front Neuroendocrinol. 30, 46–64 (2009).
- Sheehan, T. et al. Evidence that the medial amygdala projects to the anterior/ventromedial hypothalamic nuclei to inhibit maternal behavior in rats. Neuroscience 106, 341–356 (2001).
- Shahrokh, D. K. et al. Oxytocin-dopamine interactions mediate variations in maternal behavior in the rat. Endocrinology 151, 2276– 2286 (2010).
- Geary, D. C. Evolution and proximate expression of human paternal investment. Psychol. Bull. 126, 55–77 (2000).
- Fleming, A. S., Corter, C., Stallings, J. & Steiner, M. Testosterone and prolactin are associated with emotional responses to infant cries in new fathers. Horm Behav. 42(4), 399‐413 (2002).
- Abraham, E., Hendler, T., Zagoory-Sharon, O. & Feldman, R. Network integrity of the parental brain in infancy supports the development of children’s social competencies. Soc. Cogn. Affect. Neur. 11(11), 1707-1718 (2016).
- Cong, X. et al. Parental oxytocin responses during skin-to-skin contact in pre-term infants. Early Hum. Dev. 91(7), 401-406 (2015).
- Riem, M. M. et al. Oxytocin modulates amygdala, insula, and inferior frontal gyrus responses to infant crying: a randomized controlled trial. Biol. Psychiatry 70, 291-297 (2011).
- Weisman, O., Zagoory-Sharon, O. & Feldman, R. Oxytocin administration to parent enhances infant physiological and behavioral readiness for social engagement. Biol. Psychiatry 72, 982-989 (2012).
- Gettler, L. T., McDade, T. W., Agustin, S. S., Feranil, A. B. & Kuzawa, C. W. Longitudinal perspectives on fathers’ residence status, time allocation, and testosterone in the Philippines. Adapt. Hum. Behav. Physiol. 1(2), 124-149 (2015).
- Feldman, R. The adaptive human parental brain: implications for children’s social development. Trends Neurosci. 38(6), 387-399 (2015).
- Abraham, E. et al. Father’s brain is sensitive to childcare experiences. PNAS 111(27), 9792-9797 (2014).
Images:
Cover Photo from Free Images, Creative Commons CC0. https://freeimages.51microshop.com/young-boy-smiles-at-father-holding-baby-sister.html
Figure 1 created using Autodesk Sketchbook

Leave a comment