May 7, 2019
Written by: Claudia Lopez-Lloreda
Watching someone experience pain, like when watching an NBA player twist their ankle, generates a strange reaction in us: we cringe, we wince, and, occasionally, we have to look away. Even though we do not feel the physical pain in our body, we can certainly empathize with the pain others are suffering and share the emotional experience. The phenomenon where another person’s emotions trigger your own is called emotional contagion. Watching somebody else in pain specifically is called pain empathy. Evidently, there aren’t any physical changes to the body that would elicit pain, so what is happening in your brain during pain empathy?
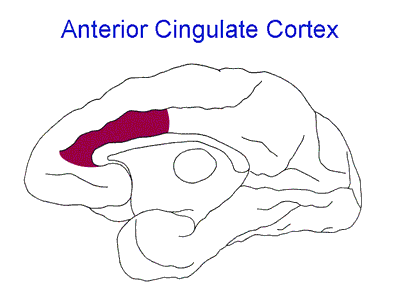
For quite some time it has been known that the anterior cingulate cortex (ACC), a region of the brain implicated in the integration of emotion and cognition, is important in the experience of pain (Figure 1). Surprisingly, when looking at pain empathy, brain imaging studies showed that the ACC was activated not only when experiencing pain but also when witnessing pain in others1. Even seeing somebody else with a facial expression of pain activates the ACC2. Additionally, reducing the activity of the ACC alters the ability to feel pain empathy3, meaning this area is important for both the experience and the observation of pain.
Activation in response to both observation and first-hand experience suggests that cells called mirror neurons may be present in the ACC. Mirror neurons are activated when an individual performs an act or experiences a sensation, as well as when a person sees another individual perform the same act or experience a similar sensation. These types of cells are believed to be essential for social interactions, as they allow us to perceive what others might be feeling and experiencing. Researchers had long hypothesized that these neurons existed for emotions, however no study had been able to precisely identify them and decipher their functional properties in any species. Therefore, a new study led by Dr. Christian Keysers at the Netherlands Institute for Neuroscience sought to find them4. They asked one important question: are there emotion-specific mirror neurons in the brain that mediate pain empathy?
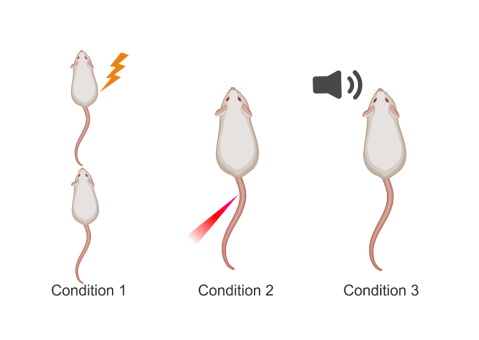
On their quest to find these neurons, the scientists used a model of emotional contagion in rats. Although empathy is a very complex human emotion, pain empathy can be evoked in rats by inducing pain in other rats (Figure 2). In this model, they measured activity of single neurons in the ACC of rats in three conditions: when seeing another rat receive a painful electric foot shock (condition 1), when being subjected to painful laser stimulation themselves (condition 2), and when hearing a tone that was previously paired with a shock (condition 3). The first condition allowed the researchers to see if neurons in the ACC responded when observing pain in others, the second to see if neurons responded to the physical experience of pain, and the third to see if neurons responded to the fear of possible painful stimuli. These conditions allowed the authors to discriminate between neurons that are responding specifically to pain (conditions 1 and 2) and other experiences and emotions such as fear (condition 3). They found that a specific subset of neurons responded both to observing another rat being shocked and to experiencing painful laser stimulus themselves. Indeed, these were the pain mirror neurons that they were looking for! Seemingly, there is a common code in our brains to process one’s own pain as well as others’. Further, they confirmed these neurons were specific to pain, as they did not respond to the tone that would cause fear, indicating that these cells were selective to a particular emotion.
Next, the researchers wanted to determine if the ACC and the mirror neurons were necessary for the behavioral response to pain empathy, or if pain empathy can still take place without them. Normally, rats tend to have a very specific behavioral response when they are distressed or experience fear: they freeze in their place. In this study, rats froze in response to both observing pain and to the fear-conditioned sound. The scientists wanted to see what happened to behavior if activity in the ACC was blocked. The researchers injected muscimol, a drug that inactivates neurons, into the ACC, where they had previously found the pain mirror neurons. Injecting this drug would effectively suppress the activity of the neurons and allow them to confirm if the ACC is needed for pain empathy. After injecting the drug, they measured the behavior of the rats in response to seeing another rat get shocked (condition 1) and hearing the fear-conditioned sound (condition 3). Interestingly, inactivating the ACC reduced freezing behavior in response to seeing another rat get shocked, but not in response to the fear-conditioned tone. This would suggest that the ACC and potentially the mirror neurons are essential in mediating socially-triggered behavioral responses as opposed to self-experienced fear.
This new study goes beyond rats. Knowing that mirror neurons for pain empathy exist in the ACC of rats now gives researchers evidence that these mirror neurons are likely to exist in humans. Studying these emotion mirror neurons could elucidate why there is decreased empathy in different conditions such as psychopathy, in which ACC activity is known to be reduced5. It could also mean something for people with brain injuries: an affected ACC may have profound impact on the ability to feel empathy for others.
Even further, the discovery of pain mirror neurons opens another possibility: the existence of other emotion-specific mirror neurons. Before this finding, most known mirror neurons were motor neurons that were activated in response to the performance and the witnessing of a task6. Pain is easier to elicit than happiness or sadness in rodents or nonhuman primates, therefore finding mirror neurons activated in response to other emotions poses a big challenge. However, the fact that these neurons exist hints at the idea that there are mirror systems in place in the brain with the purpose of interpreting the emotional experience of others. Will we find happiness mirror neurons next? The quest for more emotion mirror neurons continues…
References
- Lamm, C., Decety, J., & Singer, T. (2011). Meta-analytic evidence for common and distinct neural networks associated with directly experienced pain and empathy for pain. NeuroImage,54(3), 2492-2502. doi:10.1016/j.neuroimage.2010.10.014
- Botvinick, M., Jha, A. P., Bylsma, L. M., Fabian, S. A., Solomon, P. E., & Prkachin, K. M. (2005). Viewing facial expressions of pain engages cortical areas involved in the direct experience of pain. NeuroImage,25(1), 312-319. doi:10.1016/j.neuroimage.2004.11.043
- Mischkowski, D., Crocker, J., & Way, B. M. (2016). From painkiller to empathy killer: Acetaminophen (paracetamol) reduces empathy for pain. Social Cognitive and Affective Neuroscience,11(9), 1345-1353. doi:10.1093/scan/nsw057
- Carrillo, M., Han, Y., Migliorati, F., Liu, M., Gazzola, V., & Keysers, C. (2019). Emotional Mirror Neurons in the Rat’s Anterior Cingulate Cortex. Current Biology,29(8). doi:10.1016/j.cub.2019.03.024
- Meffert, H., Gazzola, V., Boer, J. A., Bartels, A. A., & Keysers, C. (2013). Reduced spontaneous but relatively normal deliberate vicarious representations in psychopathy. Brain,136(8), 2550-2562. doi:10.1093/brain/awt190
- Gallese, V., Fadiga, L., Fogassi, L., & Rizzolatti, G. (1996). Action recognition in the premotor cortex. Brain,119(2), 593-609. doi:10.1093/brain/119.2.593
Images
Cover image. Created with BioRender
Figure 1. From Richard Saunders, DPhil, NIMH Laboratory of Neuropsychology via Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Anterior_Cingulate_Cortex.png
Figure 2. Created with BioRender.
Hi. Can I please use your top image in a lecture to explain mirror neurons ? 🙂
LikeLike
I was also wondering if I could have permission to use your top image in a booklet on empathy development in children
LikeLike