March 3, 2020
Written by: Carolyn Keating
A tremor in the hand. Slowed movements. Stiff muscles. These are the symptoms most people associate with Parkinson’s disease. These difficulties in movement occur when neurons in a specific brain region become impaired and die1. But interestingly, research suggests Parkinson’s disease might not start in the brain at all. It turns out that the gut could be the key to the beginnings of the disease.
The gut and the brain aren’t as disconnected as they might at first seem. The two areas are able to communicate through the vagus nerve, which allows the brain to control the major organs in the body and receive sensory information about the state of those organs (Figure 1). The gut itself even has its own nervous system (called the enteric nervous system) that is so complex that it’s actually referred to as the second brain2.
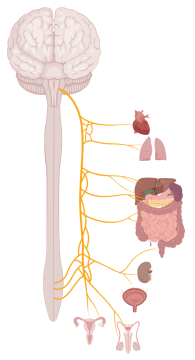
So what does the gut have to do with Parkinson’s disease? Before movement difficulties arise, gastrointestinal (GI) symptoms like constipation, nausea, and vomiting often occur first. The gut also displays some other hallmarks of Parkinson’s disease that are found in the brain. For instance, the brains of patients contain abnormal clumps of proteins called Lewy bodies. The main protein in these clumps is misfolded alpha-synuclein, and researchers believe it has an important role in causing Parkinson’s. Clumps of alpha-synuclein have been found in the enteric nervous system as well. This “sticky” alpha-synuclein can even spread from the gut, up the vagus nerve, and into the brain, where it is believed to travel between connected neurons to reach the cells responsible for controlling movement. In fact, people who have had to have their vagus nerve cut have a lower risk of developing Parkinson’s3.
While it seems that alpha-synuclein can travel from the gut to the brain, what’s not clear is how the misfolded protein affects the gut and how exactly it spreads. A new study (from a UPenn Neuroscience alum!) aimed to shed some light on what’s going on in the GI tract4. To test if gastrointestinal symptoms were caused by misfolded alpha-synuclein, the researchers injected small clumps of the protein into the small intestines of mice. Over time, the injected alpha-synuclein led to larger, toxic clumps of the protein in the gut, as well as inflammation and GI dysfunction. One way the alpha-synuclein could cause GI dysfunction is by disrupting the connectivity between cells of the enteric nervous system. And that’s exactly what the scientists saw; the enteric neurons had a reduced ability to communicate with each other.
Now that the researchers showed how alpha-synuclein could affect the gut, they wanted to see if small clumps of protein injected in the gut could make their way up to the brain, as previous studies had demonstrated. When they looked in the brain though, they didn’t see any increases in toxic alpha-synuclein, and none of the mice had any lasting movement difficulties. However, the scientists had been performing these experiments on young adult mice. Since Parkinson’s disease typically affects older people, what would happen if they used older mice? When they repeated the experiments in aged animals, the researchers again found GI tract dysfunction, but this time they also saw lasting movement difficulties. When they examined the brains of these mice, not only did they find toxic alpha-synuclein, but they also found that the neurons responsible for controlling movement weren’t functioning as they should.
What could explain the differences between the young and old mice? One possibility is that younger mice are better able to get rid of the misfolded alpha-synuclein. Cells have many proteins for “garbage disposal,” and one in particular is called glucocerebrosidase (GCase). It’s known that when GCase isn’t able to function properly, alpha-synuclein builds up. When the researchers looked at levels of GCase in the small intestine of their mice, they saw that the old animals had much less than the young animals. To test if increasing GCase could be a way to treat the clumped proteins and GI dysfunction, the scientists looked at animals that were created to have high levels of alpha-synuclein without injections into the gut. When the scientists used gene transfer to deliver the gene that makes GCase into the enteric neurons of the mice, they found higher levels of GCase and less toxic alpha-synuclein and GI dysfunction. These results suggest that treatments targeting proteins like GCase involved in “garbage disposal” could one day help patients with Parkinson’s. With more studies focused on the role of the gut, hopefully in the future doctors will be able to diagnose and treat patients before the disease progresses to debilitating movement impairments.
References
- National Institute of Aging. Parkinson’s Disease. Available at: https://www.nia.nih.gov/health/parkinsons-disease.
- Rao, M. & Gershon, M. D. The bowel and beyond: the enteric nervous system in neurological disorders. Nat. Rev. Gastroenterol. Hepatol. 13, 517–528 (2016).
- Liddle, R. A. Parkinson’s disease from the gut. Brain Res. 1693, 201–206 (2018).
- Challis, C. et al. Gut-seeded α-synuclein fibrils promote gut dysfunction and brain pathology specifically in aged mice. Nat. Neurosci. (2020). doi:10.1167/iovs.02-0285
Images:
Cover image by Wolfgang Claussen from Pixabay
Figure 1 created with BioRender

Leave a comment