February 4, 2020
Written by: Claudia Lopez-Lloreda
Many types of brain cancer exist, but one of the most threatening brain tumors are glioblastomas, malignant tumors that form when cells in the brain called glial cells divide uncontrollably. Glioblastomas account for 15% of primary brain tumors and people who suffer from glioblastoma tend to have a short average survival time of only 12-15 months. Unfortunately, glioblastoma interventions have very poor outcomes. Even the most successful interventions increase survival duration by only a mere two months.
But why? Scientists believe one of the main reasons for poor outcomes is the immense diversity of traits, such as cell type composition, that exists both across tumors in different patients and within the tumor. This makes glioblastomas particularly hard to study and treat, as each tumor seems to have a particular set of characteristics. Now, a new up-and-coming technology may help researchers understand the diversity of these malignant tumors, potentially leading to personalized treatment.
To get around the difficulty of studying the human brain, neuroscientists have developed a new model called brain organoids. These are little pea-sized replicas of the developing brain. Scientists generate the 3D structure from stem cells that turn into neurons and other cell types found in the brain. A lab led by Hongjun Song and Guo-li Ming at the University of Pennsylvania has been at the forefront of this technology.
Researchers had previously developed “cancerous” brain organoids by introducing mutations associated with tumors into organoids1 or by creating organoids from cancerous cells from patients2. However, whether these organoids were similar to the glioblastoma tumors or if they recapitulated the diversity seen across tumors was unclear.
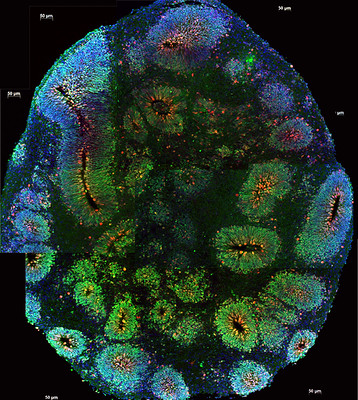
In a recent study published in Cell3, Ming, Song and colleagues generated organoids from tumors taken from glioblastoma patients to address this uncertainty. Within two weeks, pieces cut from the tumor formed into glioblastoma organoids, or GBOs, that could be kept in cell culture. Under the microscope, each GBO looked a lot like its “parental” tumor, meaning the tumor it was originally developed from.
The researchers then delved into examining just how similar the GBOs could be to their parental tumors. First, they looked at the different types of cells the GBOs were made of, particularly the number of glia, immature neurons, and neural progenitors, the cells that later give rise to neurons. GBOs had a similar diversity of cells to that seen in tumors. More importantly, the cellular composition, or the number of the different cell types, for each GBO closely resembled that of their parental tumor.
Next, the scientists looked at the gene expression profiles of GBOs and their tumors. They found that GBOs maintained the pattern of expression seen in their parental tumor over time. Since some tumors are associated with particular genetic mutations, they examined whether GBOs still had them. Even the mutations from the tumors were still present in the GBOs! This suggests that the organoids preserved the characteristics of the tumors they came from.
However, as this was all done in a dish, the researchers wanted to see if GBOs behaved the same way in animals. To do this, they transplanted intact GBOs into the brains of mice. Transplantation of GBOs in mice led to effective incorporation into the brain. Paralleling human disease, tumor cells from the GBO infiltrated the surrounding tissue, a major process seen in glioblastomas. Up to three months later, transplanted GBOs maintained important mutations present in the parental tumors.
Taking a step further, the scientists asked whether GBOs could be a model to test therapies. To test the first standard of care treatment, they treated GBOs with radiation in combination with temozolomide, a chemotherapeutic drug usually used for brain cancer. They found that some GBOs responded well to the treatment while others did not. Upon taking a closer look, researchers found that the way the GBOs responded had a lot to due with the particular mutations they had (remember that glioblastoma tumors can have different mutations). Since these mutations over-activate or inhibit different pathways, they went on to use more specific treatment to target these. They found that GBOs responded positively to treatment targeting specific pathways if they had a mutation within that pathway.
Employing another treatment, the researchers treated GBOs with CAR T-cell therapy, in which T cells, a type of immune cell, are directed to attack specific cancer cells. CAR T-cells effectively killed the cancer cells that expressed a specific type of mutation, again suggesting that clinical benefit could be mutation specific.
In this impressive study, the scientists show that organoids generated from specific glioblastoma tumors maintain the variety of characteristics at the level of cell type, gene expression, and response in brain tissue. They present an opportunity to develop a more accurate model of glioblastoma that recapitulates the diversity seen in humans and is also generated faster than other models. These glioblastoma organoids provide a lot of hope—they can serve as models to quickly study the traits of a particular glioblastoma tumor and test specialized treatment for cancer patients, hopefully leading to better outcomes for people diagnosed with glioblastoma.
References
- Bian, S., Repic, M., Guo, Z., Kavirayani, A., Burkard, T., Bagley, J. A., … Knoblich, J. A. (2018). Genetically engineered cerebral organoids model brain tumor formation. Nature Methods, 15(8), 631–639. doi: 10.1038/s41592-018-0070-7
- Hubert, C. G., Rivera, M., Spangler, L. C., Wu, Q., Mack, S. C., Prager, B. C., … Rich, J. N. (2016). A Three-Dimensional Organoid Culture System Derived from Human Glioblastomas Recapitulates the Hypoxic Gradients and Cancer Stem Cell Heterogeneity of Tumors Found In Vivo. Cancer Research, 76(8), 2465–2477. doi: 10.1158/0008-5472.can-15-2402
- Jacob, F., Salinas, R. D., Zhang, D. Y., Nguyen, P. T., Schnoll, J. G., Wong, S. Z. H., … Song, H. (2020). A Patient-Derived Glioblastoma Organoid Model and Biobank Recapitulates Inter- and Intra-tumoral Heterogeneity. Cell, 180(1). doi: 10.1016/j.cell.2019.11.036
Images
Cover image by NIH Image Gallery on Flickr, CC BY-NC 2.0.
Cool!
Love you!
Sent via the Samsung Galaxy S10e, an AT&T 5G Evolution capable smartphone
LikeLike