April 2, 2019
Written by: Rebecca Somach
People love to rank things. Everyone is guilty of spending a bit too much time online looking at top 10 lists and no one can doubt that people have a special place in their hearts for national sports championships. We love to find out what is the best of the best. Rankings aren’t just for the internet and television though, they are also found in science! Nature Methods is a scientific journal that is dedicated to publishing the latest and greatest in the way scientists collect data. Each year, the journal releases its pick for the top scientific method of the year. In 2018, the top prize went to a topic in the neuroscience field called “Imaging in Freely Behaving Animals.”1 What does that mean, why is it important, and what does it mean for the future of neuroscience?
While neuroscientists all study the nervous system, they don’t all study it in the same way. For example, if a researcher wanted to investigate why animals behave in certain ways, they could look at the way individual neurons behave. Alternatively, another neuroscientist could be examining the exact same behavior, but using whole-brain imaging. While many scientists use living cells, animals, or humans to study the nervous system, others don’t even need living tissue, but instead use computers to try to model how they think cells would communicate with each other. Why don’t these scientists just work with the brain itself if they want to know how the brain is connected to behavior? It turns out that the brain is incredibly complicated! The human brain has approximately 86 billion neurons2 and each neuron makes a connection with thousands of others. Scientists have made a lot of progress in figuring out how one single neuron can communicate with another neuron. However, adding even one or two more neurons increases the complexity of the communication. Now imagine adding thousands of other cells to this process and you’ve got an extremely confusing web of information. Right now, scientists can understand how single cells talk to each other, and they can also understand that animals behave in certain ways when put in certain situations. What they don’t yet understand is what goes on in the brain when an animal is behaving in a certain way.
There are already some options for understanding what is going on inside the brain when an animal is doing something. The human brain can be imaged using functional magnetic resonance imaging (fMRI) while the subject is answering questions or completing tasks to understand broad patterns of brain activity during certain behaviors. However, this doesn’t allow individual neurons to be investigated, not to mention being in an MRI machine isn’t a very natural situation for any human. Additionally, this technique can’t be widely applied to other animals used by neuroscientists — we can’t really ask a mouse to explain to us what they are thinking inside of an MRI machine! There are techniques to take still images of the neurons in the brain, but those types of pictures are unable to capture neuronal activity as an animal does a task. There are also techniques that are used to understand the electrical activity of neurons as animals behave, but these aren’t visual methods, so scientists cannot see which neurons are firing. The combination of both visualizing the neurons themselves and their activity in real time is what is needed to understand how individual neurons work together to influence behavior.
To image the activity of single neurons or groups of neurons, scientists can genetically create neurons that will glow when they activate (Figure 1). This glowing happens by taking advantage of the fact that when a neuron sends a signal, it needs to take in calcium. Scientists use different types of molecules that react with calcium to produce light, thus allowing them to identify which neurons are active at any moment in time based on how brightly they glow.
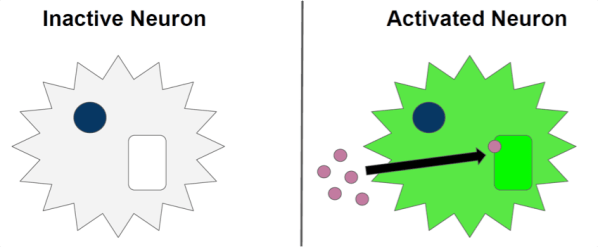
While this technique, called calcium imaging, has been around for a long time (since 19973 ), there have been limitations using it in freely moving live animals until recently. While scientists can see incredibly detailed images of cells and their connections, there have been issues with making sure the images are stable and clear. That’s because to do any of these experiments, a scientist will take an animal and attach a microscope above its brain. As you can imagine, trying to take microscopic pictures of an animal while it is moving around is not the easiest task. When camera technology was not as good as it was today, scientists could still record information from the brain but they needed to make a choice. If they wanted to observe how cells responded while an animal was freely moving, they could record the calcium levels of groups of neurons but couldn’t record them from individual neurons. Researchers could see a general increase or decrease in calcium in a brain area but there was no way to know which neurons were driving this change. If scientists did want to image individual neurons, some cameras were powerful enough but they had to be kept still. This means that to use these cameras, an animal would have to be attached to a large, immobile microscope that would not allow the animal to move around freely. Sometimes the animal would be able to walk but on a treadmill or a running wheel (Figure 2), but this is not similar to a natural situation, unless you’re studying gym rats and not actual rats.
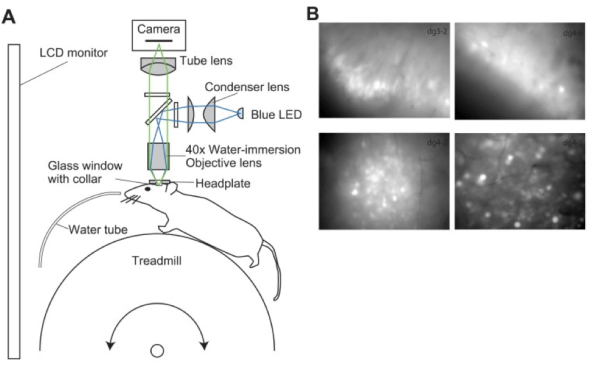
Images adapted from Hayashi et al. 20174, CC BY 4.0
When wearable cameras were developed that could actually image neurons while an animal was moving around in its normal environment, some of them were physically too heavy to be used on small animals. For instance, laboratories that study mice were unable to use the technology because the cameras would weigh down smaller mice compared to larger rats that could bear the weight.
This is where the 2018 Method of the Year comes in. It is only recently that we have the combination of lightweight, wearable cameras and genetic tools that let us take a peek into the neuronal world in real time. This means that we can now see which neurons are firing at the exact time an animal is moving around in the world, performing different behaviors! For example, the new cameras have let us see the neurons that respond when animals interact socially. In the Dulac lab in 20175, the responses of individual cells in a brain region called the amygdala were tracked as male and female mice interacted with each other and with baby mice. They saw that they could group neurons by their activity patterns in different social settings. They could also see how these neurons changed over time since they could see the same neurons for several months.
These cameras have also have let us see neurons that might be involved with forming new memories. In a study in 2013 by the Schnitzer lab6, these cameras were used to track the activity of individual neurons over several weeks. They saw that some cells change their firing patterns when an animal encounters a specific location, but other neurons fired in the same way to similar locations even after 45 days. Since these cells were so stable for so long, it is thought that their activity contributes to memory.
Now that we have the technology to see neurons communicate as an animal moves around, what can we do with that? We can not only see the neurons that are active when an animal does a particular behavior, but we can also combine that with all our other neuroscience techniques. Scientists can use genetic tools and clever behavioral tests that they have used in the past in combination with the new imaging technology to begin answering a number of questions in neuroscience that were unanswerable before. For instance, scientists want to use this technology to understand why animals might behave differently if they have different types of diseases. While we still have a long way to go with understanding the way that individual neurons and groups of neurons are involved in behavior, the 2018 Top Method of the Year is bringing us a lot closer to that goal.
Image References:
Cover image via Pixabay (https://pixabay.com/illustrations/lens-colorful-background-digital-582605/)
Figure 2 adapted from Hayashi Y, Yawata S, Funabiki K, Hikida T. In vivo calcium imaging from dentate granule cells with wide-field fluorescence microscopy. PLoS One. 2017;12(7):e0180452. Published 2017 Jul 12. doi:10.1371/journal.pone.0180452
References:
- Method of the Year 2018: Imaging in freely behaving animals, Nature Methods, 2018
- Azevedo FAC, et al. Equal numbers of neuronal and nonneuronal cells make the human brain an isometrically scaled-up primate brain. J. Comp. Neurol. 2009;513:532–541. doi: 10.1002/cne.21974.
- Miyawaki, A.; Llopis, J.; Heim, R.; McCaffery, J. M.; Adams, J. A.; Ikura, M.; Tsien, R. Y. Fluorescent indicators for Ca2+ based on green fluorescent proteins and calmodulin. Nature. 1997; 388 (6645): 882–887
- Hayashi Y, Yawata S, Funabiki K, Hikida T. In vivo calcium imaging from dentate granule cells with wide-field fluorescence microscopy. PLoS One. 2017;12(7):e0180452. Published 2017 Jul 12. doi:10.1371/journal.pone.0180452
- Li Y, Mathis A, Grewe BF, et al. Neuronal Representation of Social Information in the Medial Amygdala of Awake Behaving Mice. Cell. 2017;171(5):1176–1190.e17. doi:10.1016/j.cell.2017.10.015
- Ziv Y, Burns LD, Cocker ED, Hamel EO, Ghosh KK, Kitch LJ, El Ga-mal A, and Schnitzer MJ (2013). Long-term dynamics of CA1 hippocampal place codes. Nat. Neurosci 16, 264–266.

Leave a comment