October 5, 2021
Written by: Lisa Woodridge
A major goal of neuroscience research is to understand how neurons come together to form brain pathways and how these pathways influence behavior. An ideal way to measure this is by manipulating specific neurons and observing how other neurons or an organism’s behavior respond. However, this ideal experimental design is far from trivial.
One challenge is speed. Brain activity occurs incredibly quickly, with some neurons firing hundreds of times every second! Because of the limits of technology, many types of experimental manipulations of neurons last many, many times longer than the firing rate of a neuron. Sometimes this unnatural manipulation causes changes that would not usually happen in a normal brain, which can lead to false conclusions about how the brain really works. Another challenge is the size and organization of brain regions. Each brain region, such as the amygdala, prefrontal cortex, or hippocampus, is actually made up of many smaller groups of cells that may perform different or even opposing functions. Often, neurons that share a common function are dispersed throughout a brain region, intermingled with neurons that have different functions.
For decades, neuroscientists relied on technology such as electrical stimulation (via implanted electrodes) or drug exposure to manipulate neurons. However, these conventional techniques have important limitations. While electrical stimulation can happen incredibly quickly and thus mimic normal neuronal speed, any cell in the vicinity of an electrode will be affected by the stimulation. This could result in the manipulation of many different groups of neurons performing different functions all at once, obscuring results. Drugs can be used to target more specific groups of neurons based on the presence or absence of specific receptors. The challenge with this technique, however, is that most drugs act slowly and have effects that last for a long time – nothing at all like the normal firing rate of a neuron. These traditional methods can therefore only give limited information about the function of brain regions, pathways, and neural populations.
To overcome the limitations associated with conventional methods, neuroscientists and engineers have been developing new technology to more precisely manipulate neural activity. One such tool, called optogenetics – a method of controlling neurons with light – is revolutionizing the study of the brain.
How does optogenetics work?
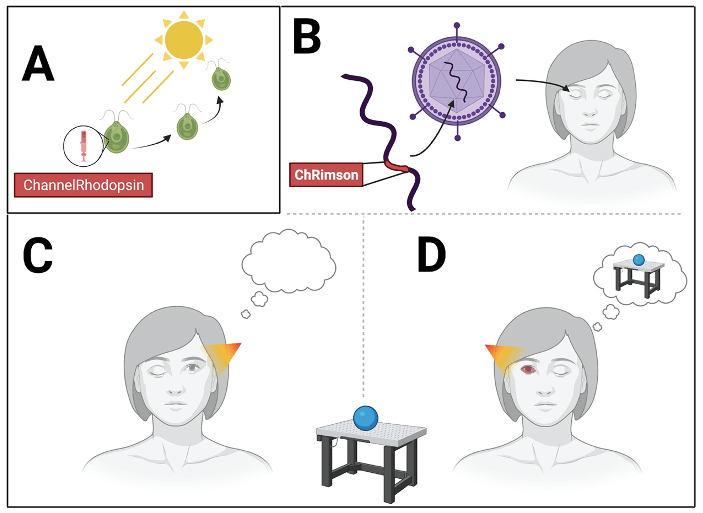
Optogenetics involves manipulating a cell’s genes (-genetics) to make it responsive to light (opto-). This is done by inserting a gene to make a specific type of protein called an opsin, into a subgroup of cells that an experimenter wants to manipulate. Opsins are found throughout nature. For example, our own eyes use rhodopsin to see the world, allowing sight to guide behaviors1. Most of the opsins used in optogenetics are derived from single-celled organisms such as bacteria and algae2, where they help control phototaxis behaviors – movement towards light to gather substrates for photosynthesis (see Figure 1A).3 Opsins work like receptors for neurotransmitters, except instead of responding to chemicals such as dopamine, they respond to beams of light! When activated by the incoming light, opsins change the electrical properties of the cell, making it more (in the case of excitatory opsins) or less (in the case of inhibitory opsins) likely the cell will fire. Neuroscientists can deliver these opsins into neurons by inserting the opsin’s gene into a virus. Since viruses proliferate by using their host cell’s gene expression machinery to replicate their own genes, using this virus delivery system means that the host cell will express not only the virus’ genes, but the opsin too! Scientists can even modify the virus to only interact with the machinery responsible for making certain host cell proteins4. Oftentimes, cells that have the same protein share a common function. For example, cells that have dopamine receptor proteins will all respond to dopamine. So by linking the virus to a specific host cell protein, opsins can be delivered selectively to groups of cells that share a similar function. This allows for functional groups of neurons to be studied in isolation, even if members of the group are interspersed throughout a brain region.
How and why is optogenetics used?
Optogenetics are widely used today in basic and translational neuroscience research. The first step to the widespread use of this technique was the discovery of the opsin Channelrhodopsin in the algae Chlamydomonas reinhardtii by Peter Hegemann and Georg Nagel in 20025. Channelrhodopsin is an excitatory opsin – when activated by light, it makes neurons more likely to fire. Since its discovery, Channelrhodopsin has been enormously consequential in understanding the role of small neural populations in the brain. Among many other discoveries, it has elucidated the functional role of different dopamine-producing neurons in the pathology of Parkinson’s disease6; clarified the precise roles of communication between the prefrontal cortex and the amygdala in the control of anxiety-related behavior7, and shown how different patterns of neural activity in a pathway between the hippocampus and the prefrontal cortex can lead to different avoidance behaviors8. The later discovery of an inhibitory ion channel called halorhodopsin in Halobacterium salinarum allowed scientists to investigate the consequences of shutting off certain pathways (in order to model certain diseases, for example)3. This inhibitory opsin now allows scientists to investigate not only the function of neurons by activating them, but also the necessity of neurons for various brain processes by inhibiting them3. Since these initial opsins were discovered, bioengineers have optimized existing opsins as well as synthesized new versions with desired features. These newly designed opsins might respond to slightly different wavelengths of light, have increased ion transport efficiency, deactivate slower or faster after light activation, and many other features that make them even more useful tools for a neuroscientist’s toolbox.9
Optogenetics offers several advantages over conventional neural manipulation technology. Using light to control cells allows for extremely fine-grain control over the timing of neural stimulation or inhibition. Because the manipulation can closely mimics a neuron’s natural activity patterns, the conclusions drawn about the function of that neuron are more likely to be sound . Additionally, viral delivery systems allow for precise targeting of specific cellular populations, targeting functional groups even when they are not clustered together. Finally, opsins all have a “favorite” color of light they respond to. For example, Channelrhodopsin responds to blue light, whereas halorhodopsin responds to amber light3. By combining opsins that respond to different color of light, scientists can simultaneously control several distinct populations of cells in the same brain.
What else can we apply optogenetics to?
The introduction of optogenetics opened new frontiers for neuroscience research. Now, the possibility that optogenetics will make it into patient care is on the horizon. The ability to activate or inactivate only a specific group of neurons with extremely precise timing is tantalizing as a possible therapy for dozens of nervous system diseases. In fact, a recent report demonstrated that optogenetics can indeed be useful in treating human diseases. In this study, researchers were interested in restoring the sight of a patient with RetinitisPigmentosa, a condition where neurons at the back of the eye that detect light gradually die10. This causes patients to progressively lose their vision. The team hoped that by infecting healthy cells with a light-sensitive opsin, they could compensate for the cells that were lost.
The team used a virus to deliver the gene for an opsin called ChRimson directly to the neurons in one of the patient’s eyes (Figure 1B). ChRimson is a modified form of Channelrhodopsin9. Like Channelrhodopsin, ChRimson is also an excitatory opsin, but while Channelrhodopsin responds to blue light, ChRimson responds to amber light. This wavelength of light is gentler on the human eye, making ChRimson more suitable for patients.
The team waited a few weeks to allow the eye’s own cells to start creating ChRimson proteins. They then used special goggles that shone amber light into the patient’s eyes. This amber light should activate the ChRimson opsin, allowing the cells of the eye to fire. To test whether the eye with the ChRimson gene could “see” better, they asked their patient to identify and grab an object sitting in front of them on a table. As expected, when asked to block the ChRimson-containing eye – testing only the vision in the untreated eye – the patient could not see whether any object had been placed on a table (Figure 1C). However, when asked to block the non-treated eye instead, not only could they detect the object, they could also reach out and grab it (Figure 1D)! Using electroencephalography (EEG) the researchers were also able to detect neural responses to the object in the visual cortex corresponding to the ChRimson-treated eye. They detected no such response in the untreated eye. This observation confirmed that the response in the visual cortex was due to the activation of the cells of the eye and not simply to light exposure. So not only did this optogenetic therapy affect the patient’s eye, but it allowed the cells to function well enough to send visual information to the brain!
The success of optogenetics to help Retinitis Pigmentosa patients in this study represents one of the greatest examples of basic scientific research informing and advancing medicine. These exciting results hopefully foreshadow the development of treatments for more Retinitis Pigmentosa patients, and for other progressive neurodegenerative conditions. Who would have thought that Drs. Hegemann and Nagel’s investigation of algae could have provided this fantastic neuroscience technology? And who can guess where the next fantastic technological leap forward will come from?
- Zhou XE, Melcher K, Xu HE. Structure and activation of rhodopsin. Acta Pharmacol Sin. 2012;33(3):291-299. doi:10.1038/aps.2011.171
- Guru A, Post RJ, Ho YY, Warden MR. Making Sense of Optogenetics. Int J Neuropsychopharmacol. 2015;18(11):pyv079. Published 2015 Jul 25. doi:10.1093/ijnp/pyv079
- Engelhard C, Chizhov I, Siebert F, Engelhard M. Microbial Halorhodopsins: Light-Driven Chloride Pumps. Chem Rev. 2018;118(21):10629-10645. doi:10.1021/acs.chemrev.7b00715
- Nayerossadat N, Maedeh T, Ali PA. Viral and nonviral delivery systems for gene delivery. Adv Biomed Res. 2012;1:27. doi:10.4103/2277-9175.98152
- Hegemann P, Nagel G. From channelrhodopsins to optogenetics. EMBO Mol Med. 2013;5(2):173-176. doi:10.1002/emmm.201202387
- Kravitz AV, Freeze BS, Parker PR, et al. Regulation of parkinsonian motor behaviours by optogenetic control of basal ganglia circuitry. Nature. 2010;466(7306):622-626. doi:10.1038/nature09159
- Adhikari A, Lerner TN, Finkelstein J, et al. Basomedial amygdala mediates top-down control of anxiety and fear. Nature. 2015;527(7577):179-185. doi:10.1038/nature15698
- Padilla-Coreano N, Canetta S, Mikofsky RM, et al. Hippocampal-Prefrontal Theta Transmission Regulates Avoidance Behavior. Neuron. 2019;104(3):601-610.e4. doi:10.1016/j.neuron.2019.08.006
- Ferenczi EA, Tan X, Huang CL. Principles of Optogenetic Methods and Their Application to Cardiac Experimental Systems. Front Physiol. 2019;10:1096. Published 2019 Sep 11. doi:10.3389/fphys.2019.01096
- Sahel JA, Boulanger-Scemama E, Pagot C, et al. Partial recovery of visual function in a blind patient after optogenetic therapy. Nat Med. 2021;27(7):1223-1229. doi:10.1038/s41591-021-01351-4
Cover image from Unsplash user Perchek Industrie
