August 3, 2021
Written by: Nitsan Goldstein
There is no question that the last 18 months have made us aware of just how easily germs can spread from one person to another. Imagine you show up for a date and the person you are with starts sniffling, coughing, and sneezing. Chances are, even before the pandemic, you’d probably want to end the night early. It turns out that avoiding social engagement with sick individuals is an evolutionarily advantageous behavior. One group of neuroscientists recently wondered if they could figure out which areas of the brain are responsible for this specific kind of avoidance.
The researchers first wanted to understand if they could use mice to identify the neural correlates of the suppression of social engagement. They placed two female mice in a cage with a male mouse and measured how often the male mounted each female. One female had been injected with a control saline solution, while the other female received an injection of lipopolysaccharide (LPS), which is a component of the cell wall of bacteria. When injected in mice, LPS causes an immune response that is similar to the response observed during a bacterial infection. The research group noted that the male mouse mounted the LPS-injected female fewer times and for shorter periods of time than he mounted the control female. Amazingly, males even avoided mounting females whose fur had just been brushed with the odor from an LPS-injected female. Together, these initial results suggest that an immune response to a bacterial infection releases a certain odor that can be sensed by male mice and cause them to mount a female significantly less often.
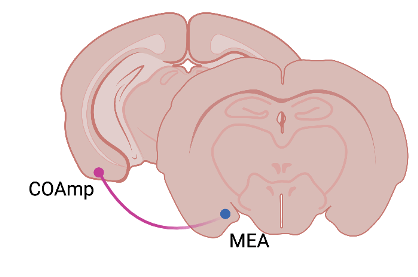
To find out which brain circuits may be responsible for this suppression in mating behavior, the group next examined regions of the brain that receive input from the accessory olfactory bulb, which is involved in recognizing socially-relevant olfactory cues. They found one area, a part of the amygdala called the COApm, which was activated in the male mice’s brains when exploring females. The activity was much higher, however, when the female had been previously injected with LPS. They also found that these neurons themselves project to another part of the amygdala, the MEA, that is also more active than normal during investigation of a sick female (Figure 1). They then turned to tools that allowed them to activate or inhibit this neural circuit and determine the effects on mounting behavior. When the scientists activated this circuit and presented males with healthy females, they mounted them less frequently. When they inhibited this circuit and presented the males with sick females, the males mounted them more often, as if they weren’t sick at all. The researchers therefore concluded that the activation of this COApm to MEA circuit is both necessary and sufficient for the suppression of mating behavior in the presence of a sick partner.
Finally, the scientists wondered if they could identify the relevant neurotransmitters and receptors involved in relaying this avoidance signal to the MEA. Using sophisticated sequencing techniques, they found that the COApm neurons that project to the MEA contain a specific hormone, Thyrotropin-releasing hormone (TRH), and that the neurons in the MEA contain the receptor for that hormone, which is called TRH receptor or TRHR. They reasoned that the release of TRH by the COApm neurons and subsequent actions on the TRH receptors in the MEA might mediate the observed effects on mating behavior. As expected, activating TRH receptors in the MEA decreased mounting behavior towards healthy females. Additionally, removing the TRH receptors from MEA neurons increased mounting of sick females.
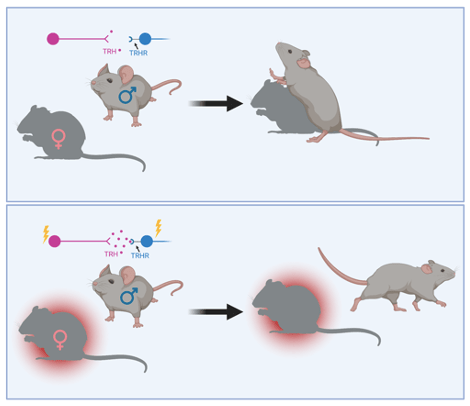
In summary, the researchers’ data suggest that a compound in the odor released by a sick female activates neurons in the COApm that contain TRH. The TRH is released onto neurons in the MEA that contain TRH receptors. Activation of these neurons interacts with other brain areas to suppress mating behavior with sick partners (Figure 2). While social interaction is important and usually beneficial for both humans and rodents, there are conditions under which it can be dangerous. It will be interesting to see whether the same neural circuit might be activated to avoid social engagement in other circumstances that make it potentially more harmful than helpful.
References:
Kwon JT, Ryu C, Lee H, Sheffield A, Fan J, Cho DH, Bigler S, Sullivan HA, Choe HK, Wickersham IR, Heiman M, Choi GB. An amygdala circuit that suppresses social engagement. Nature 593, 114-118 (2021).
Cover Image by Pixabay user Mojpe
Figures 1 and 2 created using BioRender

Leave a comment