February 23, 2021
Written by: Claudia Lopez-Lloreda
As amazing and complex as our brain cells are, they have one serious downfall: they lack the capacity to regenerate when damaged. When the axons, the long connections between neurons, of the central nervous system are severed in mammals, such as in a spinal cord injury, they fail to grow back and reconnect. This in turn can lead to movement problems and even long-term paralysis. Scientists have long sought to enhance their regenerative capacities to recover movement.
To try to help damaged neurons regenerate, most scientists have studied molecules that they would expect to improve regenerative capabilities, such as growth molecules. However, new research points to other unexpected molecules from the immune system that could help.
The capacity for neurons to regenerate is hindered by several different factors. First, when connections are severed, cells called astrocytes surround the area, forming what is called a glial scar, which blocks connections from reforming1. In addition to the astrocytes occupying the lesion, they also produce molecules called proteoglycans that inhibit growth of the axons. Proteoglycans and other inhibitory molecules flood the lesion, providing cues to stop the axons from growing. This forms a physical and chemical barrier that prevents neuronal connections from being able to reconnect.
Second, neurons lack the molecular machinery necessary to regenerate their axons. This machinery is crucial for neurons in the developing brain and spinal cord to grow, but as they mature, neurons lose this ability.
Efforts to improve regeneration either try to block the inhibitory factors that surround the lesion or improve the intrinsic capacity of neurons to be able to regenerate their connections. For example, studies have tried getting rid of pathways known to inhibit regeneration, such as a molecule called PTEN2. Other studies have tried adding in growth factors that might help regeneration. However, these have failed to recover motor function, meaning that although neurons reconnect, this does not translate into improved movement after the injury2.
Many of these have been unfruitful, and there are currently no treatments for spinal cord injury, illustrating the need for exploring new avenues to induce regeneration. This is where cytokines come in. Cytokines are molecules released by cells of the immune system that control different aspects of the immune response. Previous studies have found that certain cytokines can induce regenerative capacities of axons3. But the downside is that neurons lack or have a very low amount of the receptors necessary to be able to sense and respond to these cytokines.
To overcome this, researchers at Ruhr University of Bochum decided to introduce a hyperactive from of a cytokine, called interleukin-6 (IL-6). This means that they designed this version of the cytokine to be able to bind to a receptor expressed in most neurons so it could then activate it. Sort of like a cytokine on steroids.
In the new study published in Nature Communications, they wanted to see if this particular super cytokine could help axons regenerate after a spinal cord injury and improve functional recovery. They introduced the hyperactivated form of IL-6 after a spinal cord crush, which leads to deficits in motor function. After the injury, the mice are left to recover and researchers then examine if they have motor improvement and how well the axons reconnected, if at all.
Researchers found that 6 weeks after a spinal cord injury, mice that had been injected with the hyper IL-6 were able to regenerate their connections while control animals did not. The treatment resulted in robust regeneration of the axons beyond the crush site.
This in turn rescued the loss of motor function, allowing injured mice to walk again and engage in coordinated movement. Specifically, they found that the recovery seen was dependent on the regeneration of a specific set of axons, called the raphespinal tract (RpST). These connections originate from an area in the brain called the brainstem, whereas the hyper-IL-6 was injected into another area called the sensorimotor cortex. Interestingly, they found that IL-6 was being released from the axons that originate at the sensorimotor cortex into the connections with the raphespinal tract axons.
Although this was just done in rodents, it reveals an important mechanism that is critical in inducing regeneration. It also paves the way to explore these immune molecules as potential therapeutic treatment for spinal cord injury in humans.
Images
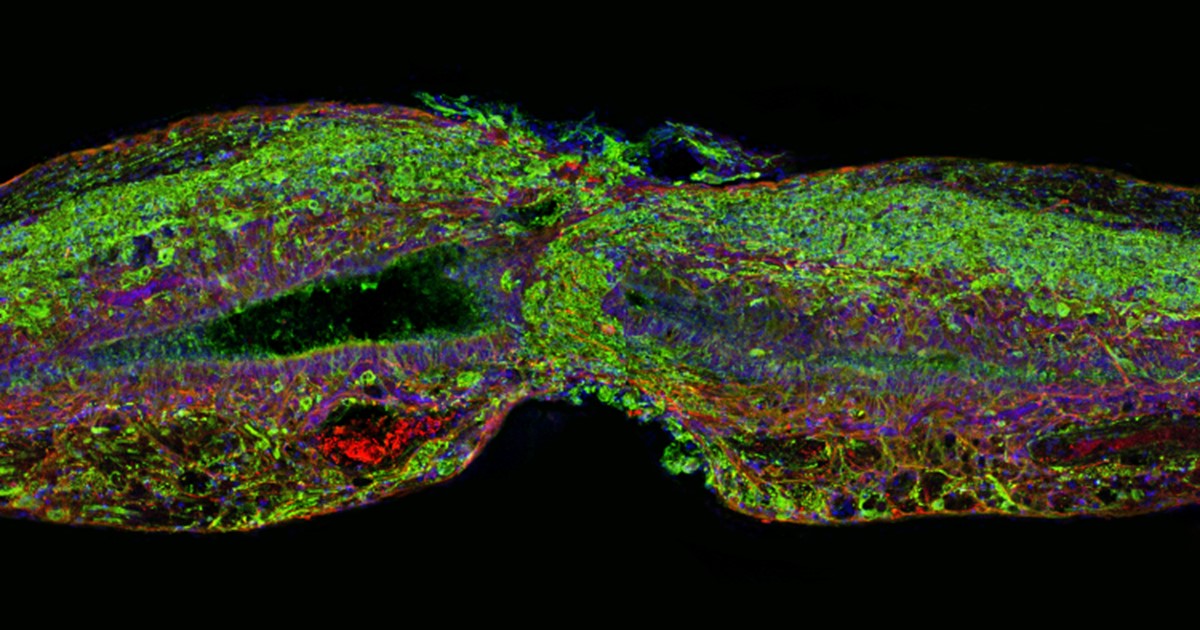
Cover image. Zebrafish spinal cord regenerating after injury. Glial cells (red) cross the gap between the severed ends followed by neuronal cells (green). Cell nuclei are stained blue and purple. Credit: Mayssa Mokalled and Kenneth Poss, Duke University, Durham, NC. From Flickr.
References
- Silver, J., & Miller, J. H. (2004). Regeneration beyond the glial scar. Nature Reviews Neuroscience, 5(2), 146–156.
- Geoffroy, C. G., Lorenzana, A. O., Kwan, J. P., Lin, K., Ghassemi, O., Ma, A., … Zheng, B. (2015). Effects of PTEN and Nogo Codeletion on Corticospinal Axon Sprouting and Regeneration in Mice. Journal of Neuroscience, 35(16), 6413–6428.
- Leibinger, M., Muller, A., Andreadaki, A., Hauk, T. G., Kirsch, M., & Fischer, D. (2009). Neuroprotective and Axon Growth-Promoting Effects following Inflammatory Stimulation on Mature Retinal Ganglion Cells in Mice Depend on Ciliary Neurotrophic Factor and Leukemia Inhibitory Factor. Journal of Neuroscience, 29(45), 14334–14341.
- Leibinger, M., Zeitler, C., Gobrecht, P., Andreadaki, A., Gisselmann, G., & Fischer, D. (2021). Transneuronal delivery of hyper-interleukin-6 enables functional recovery after severe spinal cord injury in mice. Nature Communications, 12(1).
Leave a comment