September 11, 2018
Written by: Nitsan Goldstein
What does “epigenetics” mean?
“Epigenetics is a useful word if you don’t know what’s going on – if you do, you use something else.” –Adrian Bird
In this quote Adrian Bird perfectly describes the word “epigenetics.” It means everything, therefore it means nothing. It’s an umbrella term; a word that was first used in the 1940s and 1950s when very little was known about DNA and inheritance to describe phenomena that fell outside the boundaries of classically described patterns1. So, what do people mean when they say “epigenetic,” and how does it relate to our brain development?
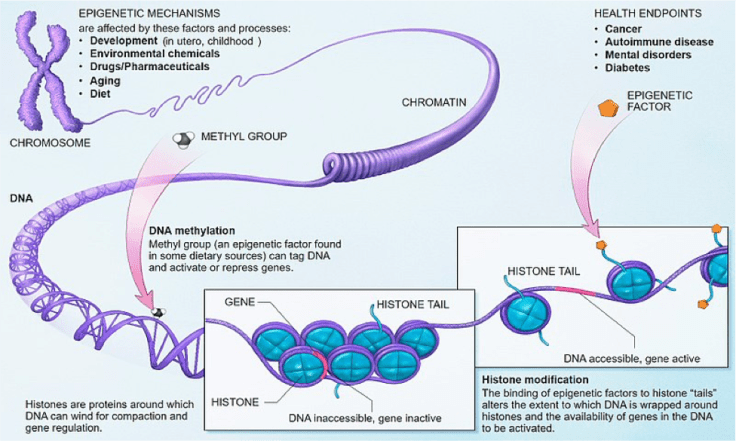
You may not have heard the term “epigenetics,” but you likely have heard the term “genetics” or “genes.” Genes are segments of DNA that form the blueprint a cell needs to make proteins. During sexual reproduction, a sperm carrying one copy of paternal DNA and egg cell carrying one copy of maternal DNA combine to form an embryo. That embryo therefore contains two copies of each gene, one from the mother and one from the father. That single cell then divides to eventually form every cell in the body, each of which contains that same DNA that was in the egg and the sperm. Some genes create proteins that affect traits like hair and eye color, which is the reason people often resemble their parents and siblings. The passing on of genes from parent to child in the form of DNA is called classical inheritance. However, it became clear early on that the DNA blueprint alone cannot contain all the information cells need to function. This is where epigenetics comes in. Epigenetics refers to factors that determine which genes are expressed, or used, by a cell that are outside the DNA blueprint. Some of these factors are themselves heritable, or passed down to one’s offspring. Think of DNA as the words used in a sentence, and epigenetics as body language and intonation. The same sentence can have various meanings if delivered differently. For more details on epigenetic mechanisms and how they affect DNA, see Figure 1.
What makes a neuron a neuron?
Remember, all cells in the body contain the exact same DNA. But think about how many different cell types a human body has. We have hair cells, skin cells, kidney cells, liver cells, neurons, and many many more. How can the same set of genes instruct such a wide variety of cell types? It turns out that while all cells contain every gene, they are not all used. Each gene is either on or off, meaning it is either expressed or not expressed. Neurons become neurons when the genes necessary to function as a neuron, such as those that code for the production of neurotransmitters or certain ion channels, are turned on, and those that are important for skin cells, for example, are turned off. Special proteins, called methyltransferases, are able to turn genes off by adding compounds called methyl groups onto DNA, which physically block that region from being expressed2. By blocking non-neuron genes and leaving neuron genes free, methyltransferases, through an epigenetic mechanism, assist in differentiating neurons and other cell types to allow them to carry out their specific functions. In this way, epigenetic modifications are very important for proper brain development. Next, we’ll focus on a rare but important kind of DNA methylation called genomic imprinting.
Genomic imprinting and brain development
In over 99% of genes, expression does not depend on whether that gene copy came from the mother or father. However, a rare epigenetic phenomenon, called genomic imprinting, occurs when groups of genes are systematically turned on or off such that only the paternal or maternal copy are expressed. There are several theories about why genomic imprinting may have evolved. It is thought, for example, that certain paternally expressed genes aid in offspring suckling behavior, giving that litter an advantage over others and therefore helping the father to ensure the passing on of his genes to future generations3.

Random mutations in DNA can often shed light on the importance of epigenetic modifications like genomic imprinting. Let’s take chromosome 15 for example. Normally, DNA methylation silences some genes in this region that are on the paternal chromosome 15, and other regions that are on the maternal chromosome 15. A rare random mutation can cause a part of chromosome 15 to be deleted in either sperm or egg cells. This mutation causes the offspring to suffer from a neurological disorder, but because these genes are imprinted, the origin of the mutation determines the outcome. If the mutation came from the father’s sperm, the result is Prader-Willi Syndrome, but if the mutation came from the mother’s egg, the result is Angelman’s Syndrome (see Figure 2). Prader-Willi Syndrome is characterized by severe overeating, potentially due to low activity in brain regions involved in the signaling of satiety4. Angelman’s Syndrome, on the other hand, is characterized by intellectual disability, developmental disability, and seizures. These two dramatically different diseases arise from a deletion of the same region of the same chromosome. However, because the paternal copies of some genes and the maternal copies of others are typically silent, a missing paternal copy and missing maternal copy have drastically different effects.
Epigenetics as a therapeutic tool
These diseases highlight the important role that epigenetic modifications to our genes play in typical brain development and function. As we learn more about the normal role of epigenetics, scientists can now attempt to develop epigenetic strategies to treat various neurological conditions. For instance, neurons that are lost or damaged in spinal cord injuries or neurodegenerative diseases could potentially be replaced by using epigenetics to reprogram a person’s skin cells into new neurons5. This immense therapeutic potential has motivated research into epigenetic mechanisms that will continue to expand our knowledge in the coming years.
Image References:
Cover photo from Wikimedia Commons, attributed to Zephyris at English language Wikipedia CC BY-SA 3.0. https://commons.wikimedia.org/wiki/File:Nucleosome_1KX5_2.png
Figure 1 from the National Institutes of Health https://commonfund.nih.gov/epigenomics/figure
References:
- Ledford H. Language: Disputed Definitions. 2008 (7216):1023-8.
- Reik W, Dean W, & Walter J. Epigenetic Reprogramming in Mammalian Development. Science. 2001 (5532):1089-93.
- Wilkinson, L. S., Davies, W. & Isles, A. R. Genomic imprinting effects on brain development and function. Nat. Rev. Neurosci. 2007 (8)832.
- Shapira NA, Lessig MC, He AG, et al.Satiety dysfunction in Prader-Willi syndrome demonstrated by fMRI. Journal of Neurology, Neurosurgery & Psychiatry 2005 (76)260-262.
- York, E.M., Petit, A. & Roskams, A.J. Epigenetics of Neural Repair Following Spinal Cord Injury. Neurotherapeutics2013 (10)757.