August 9th, 2022
Written by: Kara McGaughey
When’s the last time you used a map? Was it your GPS on the way to work this morning? The doppler radar when you checked the weather? Whatever their function, maps are ubiquitous in everyday life as intuitive ways to represent and organize the world around us.
The brain uses maps to represent and organize information, too. Just as we create different maps to highlight and interact with different features of our physical world (e.g., highways, landscapes, etc.) the brain creates maps with different features of our physical experience (e.g., touch, smell, sound, visual space, etc.). These maps enable similar information to be stored in similar places, making it easier for the brain to keep track of what’s going on and guide our behavior accordingly.
How does the brain use maps to represent our body?
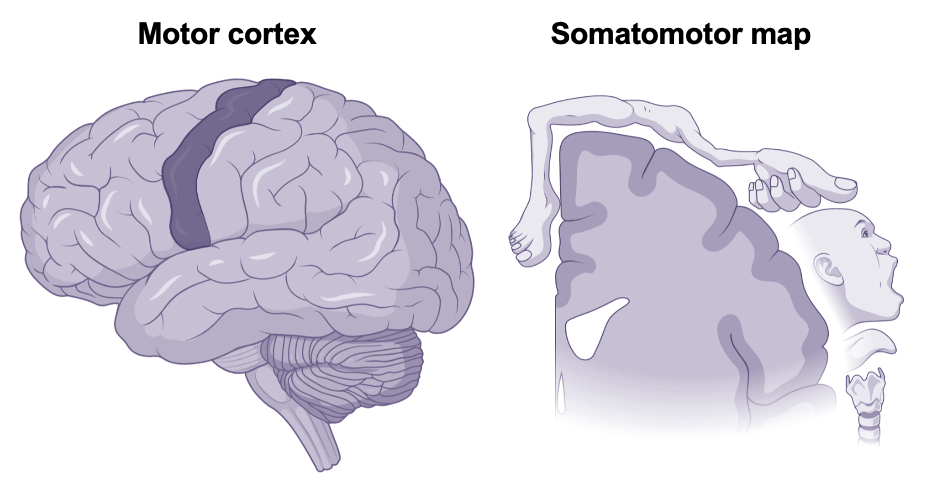
You might keep your maps half-heartedly folded in your car’s glove box, but the brain smooths its maps out across the cortical surface. One of the brain’s most well-known cortical maps forms a vertical strip that originates behind the ears and stretches toward the top of the head. This strip of brain tissue contains our motor cortex with its somatomotor map (See Figure 1). At first glance, something about this somatomotor map seems a bit off. Not only does the body appear upside down, but the relative size of the body parts is extremely distorted. (Can you imagine having a face or a hand larger than your torso?!) This distortion comes from the fact that the amount of space within the cortical map devoted to any given body part is not proportional to that part’s size, but rather to the complexity of its motor capabilities. In other words, areas of the body with more precise motor control — like our fingers — take up more space.
How does our experience change these maps?
The brain’s maps are precisely curated, but they’re not fixed. Just like how GoogleMaps continually updates and reorganizes as new information is received, the brain’s maps can change and rearrange. Brain maps can update as we learn new things or following damage to the brain or body. (You can read more about how learning shapes motor cortex in this recent PennNeuroKnow article). As one example, somatomotor maps of professional violin players who make remarkably precise string manipulations and bow movements have much larger representations of the fingers than the average adult.1 This redesignation of cortical surface area reflects the fact that the brain is continually optimizing for (e.g., prioritizing) movements or experiences that occur frequently. The reverse is also true. If there’s prolonged disuse or amputation of a body part, the brain reorganizes by giving away its cortical space.2 If, for instance, you had your thumb removed, the cortical representation from neighboring areas (in this case the forehead and the index finger) would start to take over what used to be the thumb’s piece of the map. These changes in how the brain maps and organizes information are referred to as plasticity.
While there have been a host of experiments exploring how use and disuse of particular body parts cause changes in motor maps, it wasn’t until recently that neuroscientists took a deeper dive into how exactly (and how quickly) these plasticity-related changes occur and what they might mean for how the brain functions.
How do cortical maps change and rearrange?
Exploring how the brain’s cortical maps change and rearrange involves inducing plasticity in human subjects while repeatedly measuring their brain activity over time. In a recent study, Dillan Newbold and colleagues at Washington University School of Medicine in St. Louis did just this — inducing disuse-related plasticity by putting participants’ dominant arm into a cast and monitoring their brain activity using daily 30-minute functional magnetic resonance imaging (fMRI) scans.3 (Over the course of the ~6 week experiment, this amounted to more than 150 scans!) Unlike traditional fMRI scans where participants perform a particular task inside the scanner, Dillan and colleagues took a different approach. By measuring the brain’s spontaneous activity as subjects simply rested in the scanner, they were able to get a more generalizable picture of brain activity.
When looking at this resting-state brain activity, researchers found something interesting: large pulses of spontaneous activity that appeared after casting in regions of the somatomotor map corresponding to the disused arm. The pulses propagated (or spread) from upper-arm-specific areas through the rest of the motor cortex. Discovering these large, spontaneous pulses in fully-developed adult brains was surprising because they’re usually thought of as a hallmark feature of development. Previous work, for example, has linked pulses of spontaneous activity to the establishment of brain circuits in fetal brains and identified their propagation as responsible for the initial mapping and organization of the visual system (even before the eyes have opened!).4,5
The presence of these pulses during the casting period provided evidence that participants’ brains were reorganizing in response to upper extremity disuse. When the brain stopped receiving motor information from the casted arm, it seemed to turn on these pulses of spontaneous activity as a signal that priorities had changed and cortex should remap accordingly.
What does this mean for brain function?
At the same time that Dillan and colleagues found pulses of spontaneous activity in somatomotor regions for the disused arm, they also found changes in participants’ brain function. Using a popular technique called functional connectivity that allowed them to determine the strength of connections between different parts of the brain, researchers identified a weakening of connections between regions of the somatomotor map corresponding to the casted upper extremity and the rest of the motor cortex (Figure 2, left and center). Over the course of the casting period, the functional connectivity of casted upper extremity regions creeped as low as has been observed after motor stroke or limb amputation. In other words, just 2 weeks of dominant arm disuse was sufficient to cause upper extremity somatomotor areas to basically stop communicating.

How long does it take for things to remap?
Given the substantial remapping and reorganization caused by dominant arm disuse, Dillan and colleagues were shocked to find how quickly these plasticity-related changes took place. Changes in the brain’s functional connectivity emerged roughly 48 hours after casting, suggesting that the disconnection of the disused arm from the rest of motor cortex happened on the order of days. Remarkably, just as quickly and completely as disuse of the arm had caused disconnection, reintroducing its use caused reconnection. Once the casting period was over, the pulses of spontaneous activity subsided and the disused somatomotor regions reformed their connections with the rest of the motor cortex, restoring functional connectivity between brain regions to what it looked like before the casts were put on (Figure 2, right).
This rapid reorganization highlights the brain’s remarkable capacity for plasticity and underscores just how much our recent experience can impact brain maps and brain function. So, the next time you or someone you know ends up in a cast or a boot, take a moment to appreciate that as the way they interact with the world changes, the way their brain represents and responds to the world changes, too.
References
1. Elbert T., Pantev C., Wienbruch C., Rockstroh B., Taub E. (1995). Increased Cortical Representation of the Fingers of the Left Hand in String Players. Science, 270, 305-307. https://www.jstor.org/stable/2888544
2. Mersenich M. M., Nelson R. J., Stryker M. P., Cynader M. S., Schoppmann A., Zook J.M. (1984). Somatosensory cortical map changes following digit amputation in adult monkeys. Journal of Comparative Neurology, 222(4), 591-605. https://doi.org/10.1002/cne.902240408
3. Newbold D. J., Laumann T. O., Hoyt C. R., …, Petersen S. E., Snyder A. Z., Dosenbach N. U. F. (2020). Plasticity and Spontaneous Activity Pulses in Disused Human Brain Circuits. Neuron, 107, 580–589. https://doi.org/10.1016/j.neuron.2020.05.007
4. Tolonen, M., Palva, J.M., Andersson, S., and Vanhatalo, S. (2007). Development of the spontaneous activity transients and ongoing cortical activity in human preterm babies. Neuroscience, 145, 997–1006. https://doi.org/10.1016/j.neuroscience.2006.12.070
5. Ackman, J.B., Burbridge, T.J., and Crair, M.C. (2012). Retinal waves coordinate patterned activity throughout the developing visual system. Nature,490, 219–225. https://doi.org/10.1038/nature11529
Cover photo by mohamed_hassan on Pixabay