July 14, 2020
Written by: Carolyn Keating
It’s summertime, which means people are spending more time outdoors. Wooded areas can be a great place to escape the heat, but they also pose a danger in the form of blood-sucking ticks. When these insects bite humans, they can pass on disease-causing bacteria. Certain species of ticks in the northeastern and midwestern United States and parts of Europe and Asia can pass on strains of the bacterium Borrelia burgdorferi, which causes Lyme disease. Lyme borreliosis, as the disease is formally known, was first recognized in 1976 due to a cluster of children in Lyme, Connecticut with arthritis. It was later discovered that this arthritis was just one symptom of a complicated disease that can affect the skin, joints, heart, and nervous system.1
How exactly does this bacteria get in the body and cause so many different symptoms? Borrelia burgdorferi uses deer or black-legged ticks as vectors to pass between vertebrate hosts. In June and July when tiny (and we’re talking poppy seed tiny) infected nymphal ticks are looking for mammals to feed on, the bacteria can spread to humans after about 36 hours of tick attachment. Once a tick deposits the bacteria in the skin, the bacteria multiply at the site of the bite before spreading throughout various tissues, where they have a few tricks for evading the immune system. Eventually however, both the innate and adaptive immune system are able to kill the foreign invader. It is actually this immune response that causes the symptoms of Lyme disease, since the bacterium itself does not produce any toxins.1
There are three stages of Lyme borreliosis. Stage 1 begins during the summer and usually presents several days after the infection with an expanding rash at the site of the bite called erythema migrans (Figure 1). However, not all people will develop a rash. In the US but not Europe (where the sub-species of disease-causing bacteria are different than in the US), this rash is usually accompanied by general malaise, headache, joint pain, muscle aches, fever, and swollen lymph nodes.1
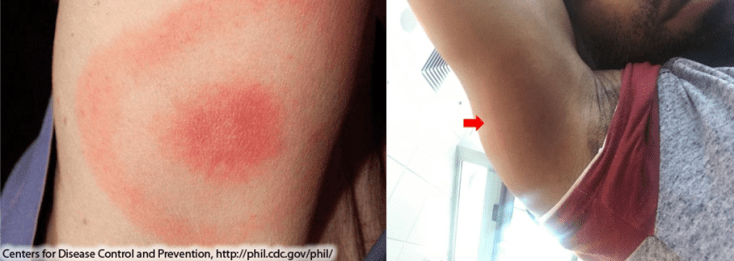
Within days to weeks, the bacteria spread from the site of the bite during early disseminated infection (stage 2). In addition to spreading to other areas of skin and causing more rashes, it can also spread to other organs. In the heart for instance, electrical impulses from the atria to the ventricles can be interrupted in what’s known as atrioventricular nodal block.1 In about 15% of untreated patients the infection spreads to the nervous system as acute Lyme neuroborreliosis.2
In the US, Lyme neuroborreliosis usually involves inflammation of the meninges (lymphocytic meningitis), the membranes separating the brain and spinal cord from bone, resulting in occasional headaches and mild neck stiffness; cranial nerve damage (neuropathy), especially to the nerve innervating the face, causing weakness of the facial muscles (facial palsy); or inflammation where sensory or motor peripheral nerves enter/exit the spinal cord (radiculoneuritis), producing pain that radiates from the back and hip into the legs that can be accompanied by numbness, tingling, and muscle weakness or heaviness. In Europe, similar symptoms are seen called Bannwarth syndrome.1,2
To produce these symptoms, the bacteria likely travel through the blood (though possibly along peripheral nerves) to the brain and spinal cord; however it is unclear exactly how they cross the blood-brain barrier to gain access to the central nervous system. Once inside, the bacteria encounter local immune cells, which secrete pro-inflammatory molecules and molecules to attract even more immune cells to the site, particularly antibody-producing B cells as well as cytotoxic T cells. There are a few ways that the bacteria may injure neurons in the central nervous system. Immune cells activated in response to the bacteria as well as glia can secrete molecules that injure neurons. The neurons could also be damaged from the bacteria themselves or through autoimmune reactions, when antibodies to the bacteria also react to structures on neurons, causing the immune system to destroy healthy neurons.3 Damage to neurons or their axons may explain the neurological symptoms of Lyme disease.
Late infection (stage 3) is typically localized to certain regions of the body, unlike the systemic responses in stage 2. At this stage months to years after infection, arthritis in one or a few joints, particularly the knee, is common in the US. In Europe, a skin condition affecting distal parts of the limbs called acrodermatitis chronica atrophicans is more frequent. Rarely, neurological symptoms such as stroke-like symptoms, cognitive impairment, or neuropathy are seen late in disease, though the existence of these syndromes is controversial.1
Also controversial is the term “chronic Lyme disease.” About 10% of patients with erythema migrans and maybe even more patients with Lyme neuroborreliosis, report symptoms such as fatigue, cognitive impairment, and musculoskeletal pain at least 6 months after antibiotic treatment that can persist intermittently for over 10 years in some cases! Lyme neuroborreliosis patients can also experience long-term motor deficits, and rarely can develop autoimmune diseases. These post-treatment symptoms often improve within months, but sometimes can persist for years. In a small proportion of patients, these symptoms interfere with daily functioning and are referred to as post-treatment Lyme disease syndrome (PTLDS), sometimes called chronic Lyme disease.1 However, the existence of PTLDS is controversial because the symptoms and underlying causes may not be the same in all patients, the syndrome has not been associated with infections in the brain and spinal cord, and because chronic Lyme disease has become a diagnosis for medically unexplained symptoms even in patients with no evidence of past or present Lyme disease.1,4
Luckily, in most patients Lyme disease is easily treated with antibiotics (and usually improves or resolves without treatment, albeit more slowly) and most patients make a full recovery once diagnosed. Diagnosis, which is confirmed by presence of a bullseye rash or through blood testing,1 is complicated this year by the novel coronavirus. You may have noticed that some of the early symptoms of Lyme disease—fever, fatigue, headache, and muscle aches—resemble those of COVID-19. Due to these similarities, it’s more important than ever this season to monitor yourself for symptoms and use precautions to prevent tick bites. Avoiding of areas with high numbers of ticks, wearing protective clothing, using tick repellent, and checking for the presence of ticks on yourself after you’ve been outdoors are all easy steps to avoid tick bites and Lyme disease.1
References:
- Steere AC, Strle F, Wormser GP, et al. Lyme borreliosis. Nat Rev Dis Prim. 2016;2:1-18. doi:10.1038/nrdp.2016.90
- Marques AR. Lyme Neuroborreliosis. Continuum (N Y). 2015;21(6):1729-1744.
- Rupprecht TA, Koedel U, Fingerle V, Pfister H. The Pathogenesis of Lyme Neuroborreliosis : From Infection to Inflammation. Mol Med. 2008;14(3-4):205-212. doi:10.2119/2007-00091.Rupprecht
- Halperin JJ. Lyme neuroborreliosis. Curr Opin Infect Dis. 2019;32(3):259-264. doi:10.1097/QCO.0000000000000545
Images:
Cover image via Elionas from Pixabay.
Figure 1: Left via Centers for Disease Control and Prevention, https://www.cdc.gov/lyme/signs_symptoms/rashes.html. Right via Dennison R, Novak C, Rebman A, Venkatesan A, Aucott J. Lyme Disease with Erythema Migrans and Seventh Nerve Palsy in an African-American Man. Cureus. 2019;11(12):e6509. doi:10.7759/cureus.6509. CC-BY 3.0.
