November 20, 2018
Written by: Carolyn Keating
Imagine that after not being able to move your legs for years, you’re able to walk again, possibly without any assistance. Sounds too good to be true, right? Well just three weeks ago a research group from Switzerland reported that that’s exactly what happened for three patients with spinal cord injury, thanks to a revolutionary new way of using a spinal cord implant.
How does paralysis happen in the first place?
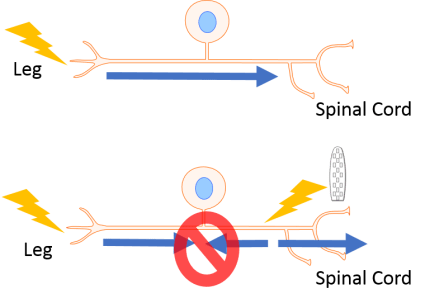
During a spinal cord injury (SCI), the communication between the brain and the nervous system below the injury is disrupted. In the case of injuries that produce leg paralysis, that means that signals from the brain telling the legs to move can’t get through, and signals from the legs carrying information about touch and limb position don’t get back to the brain (Figure 1, left).

While the communication between the brain and the circuits past the injury is severed, the circuits themselves aren’t actually damaged. Networks of neurons in the spinal cord itself can actually generate complex movements without input from the brain; they just have no way of receiving information about when or how to produce movement. That’s where epidural electrical stimulation (EES) comes in. In a lower part of the spinal cord called the lumbar region, a network of neurons forms a central pattern generator that produces rhythmic motor behavior like walking and running. After SCI, this network is “silent” as it is no longer receiving enough input from higher centers due to the injury. But placing stimulating electrodes over this region can “wake up” this network of neurons so that residual connections from the brain, as well as sensory information from the limbs, can become a signal to produce movement1 (Figure 1, right).
EES technology itself isn’t new; in fact, it’s been around for decades. And in animal models of SCI, it has been very successful: in these experiments, movement can be readily restored to the point that they can walk without any stimulation2. However, the story has been different in humans. Until recently EES has only been able to produce isolated leg movements after a year of intense rehabilitation and only while the stimulation is still on3. This September though, two groups showed that constant EES—along with extensive rehabilitation before and after stimulation—did enable several patients with complete paralysis to regain the ability to walk with assistive devices4,5. But if this technology works so well in animals, why has it been having trouble translating into people?
How is human paralysis different?
To understand why EES works differently in animals such as rats than in humans, we first need to think about how neurons normally fire. When a neuron communicates with another neuron, it generates an electrical signal called an action potential that starts at one end of the neuron and travels all the way down its long axon to its target. Importantly, this signal only travels in one direction (Figure 2, top). In contrast to normal neuron firing, EES doesn’t stimulate the part of the neuron where action potentials normally start. Instead it starts somewhere along the axon, meaning that not only can the electrical signal travel down the axon towards its target, but it can also travel backwards. This backwards action potential can crash into a natural action potential that started at the cell body and is travelling the correct direction, cancelling it out (Figure 2, bottom).
It turns out that because humans are bigger than rats and have longer, larger axons, these forwards and backwards action potential collisions during EES happen much more often in humans than in rodents6. And the signal that gets cancelled out is carrying information from the body to the brain and spinal cord about where the body is in space and how it’s oriented. In addition to providing conscious perception of leg position, this information is normally used to make sure the right muscle groups are activated to produce movement. In order for this proprioceptive information to get through, only a narrow range of EES stimulation can be used, which doesn’t really enable meaningful improvements in humans.
Scientists thought there might be a work around. In previous trials of EES, only continuous stimulation was used. But what if they delivered EES stimulation in bursts?
Non-continuous EES enables walking
What if stimulation was only delivered to neurons controlling certain muscle groups at specific times during the process of walking? To test this idea, researchers recruited three patients who sustained an SCI over four years ago and had either permanent motor deficits or complete leg paralysis. They implanted an EES device over the regions of the spinal cord that control muscles from the hips to the ankles (personalized for each patient). A pulse generator that enabled the EES to deliver the stimulation (essentially a power source/battery) was implanted in each patient’s abdomen. This pulse generator was controlled wirelessly through a programmer worn on a belt that received information from a nearby computer. Patients were suspended in a gravity-assist device that supported their body weight, and researchers wirelessly recorded their kinematics, leg muscle electrical activity, and ground reaction forces as they attempted to walk. In this way, researchers could program the EES to deliver stimulation to nerves controlling different muscle groups at different times based on how exactly the patient was trying to move.
After just five days of patients regulating the timing of their movements to pre-programmed EES sequences designed from healthy people walking, patients were able to walk on a treadmill using gravity-assist while the location/timing-specific EES stimulation was delivered. After five months of EES-enabled rehabilitation 4-5 times per week, 2 of the 3 patients regained the ability to walk without crutches or hip support, with only 35% of their body weight supported by the gravity-assist. Even without the EES on, patients regained voluntary leg movements, and could transition from sitting to standing and walking with crutches (or in the case of one patient, even walk several steps completely unassisted!). Importantly, the patients’ muscles only moved when they wanted them to—they were the ones in control7. (Check out the amazing videos of patients walking again here: https://www.youtube.com/watch?v=XFXWR4b9iVA)
This remarkable innovation has accomplished what was once unthinkable, and enabled people with leg paralysis to walk again. Although all participants still required minimal assistance to walk, it is a huge step forward (no pun intended). Researchers are already hard at work translating their laboratory set-up into something that can be used in the real world: wearable kinematic trackers on the ankles can send information to a tablet that adjusts EES stimulation in real time to the intended movement. While it’s unclear if this technology will work as well for patients who have no residual motor abilities, researchers are optimistic that one day it can be widely used to help many people with SCI.
References:
- Moraud, E. M. et al. Mechanisms Underlying the Neuromodulation of Spinal Circuits for Correcting Gait and Balance Deficits after Spinal Cord Injury. Neuron 89, 814–828 (2016).
- Shah, P. K. & Lavrov, I. Spinal Epidural Stimulation Strategies: Clinical Implications of Locomotor Studies in Spinal Rats. Neuroscientist 23, 664–680 (2017).
- Angeli, C. A., Edgerton, V. R., Gerasimenko, Y. P. & Harkema, S. J. Altering spinal cord excitability enables voluntary movements after chronic complete paralysis in humans. Brain 137, 1394–1409 (2014).
- Angeli, C. A. et al. Recovery of Over-Ground Walking after Chronic Motor Complete Spinal Cord Injury. N. Engl. J. Med. 379, 1244–1250 (2018).
- Gill, M. L. et al. Neuromodulation of lumbosacral spinal networks enables independent stepping after complete paraplegia. Nat. Med. 24, 1677–1682 (2018).
- Formento, E. et al. Electrical spinal cord stimulation must preserve proprioception to enable locomotion in humans with spinal cord injury. Nat. Neurosci. (2018). doi:10.1038/s41593-018-0262-6
- Wagner, F. B. et al. Targeted neurotechnology restores walking in humans with spinal cord injury. Nature 563, 65–71 (2018).
Image References:
Cover image: David Mzee, Wings For Life World Run 2019; courtesy of Wings For Life Spinal Cord Research Foundation via Red Bull Content Pool. https://image.redbull.com/rbx00516/0010/1/478/300/451/fileadmin/user_upload/images/DavidM_EPFL_composition__c_EPFL_Hillary_Sanctuary.jpg
Figure 1: Adapted from BruceBlaus via Wikimedia Commons, CC BY 3.0. https://commons.wikimedia.org/wiki/File:Blausen_0822_SpinalCord.png

Leave a comment